+検索条件
-Structure paper
| タイトル | Structure of the nucleotide exchange factor eIF2B reveals mechanism of memory-enhancing molecule. |
|---|---|
| ジャーナル・号・ページ | Science, Vol. 359, Issue 6383, Year 2018 |
| 掲載日 | 2018年3月30日 |
 著者 著者 | Jordan C Tsai / Lakshmi E Miller-Vedam / Aditya A Anand / Priyadarshini Jaishankar / Henry C Nguyen / Adam R Renslo / Adam Frost / Peter Walter /  |
| PubMed 要旨 | Regulation by the integrated stress response (ISR) converges on the phosphorylation of translation initiation factor eIF2 in response to a variety of stresses. Phosphorylation converts eIF2 from a ...Regulation by the integrated stress response (ISR) converges on the phosphorylation of translation initiation factor eIF2 in response to a variety of stresses. Phosphorylation converts eIF2 from a substrate to a competitive inhibitor of its dedicated guanine nucleotide exchange factor, eIF2B, thereby inhibiting translation. ISRIB, a drug-like eIF2B activator, reverses the effects of eIF2 phosphorylation, and in rodents it enhances cognition and corrects cognitive deficits after brain injury. To determine its mechanism of action, we solved an atomic-resolution structure of ISRIB bound in a deep cleft within decameric human eIF2B by cryo-electron microscopy. Formation of fully active, decameric eIF2B holoenzyme depended on the assembly of two identical tetrameric subcomplexes, and ISRIB promoted this step by cross-bridging a central symmetry interface. Thus, regulation of eIF2B assembly emerges as a rheostat for eIF2B activity that tunes translation during the ISR and that can be further modulated by ISRIB. |
 リンク リンク |  Science / Science /  PubMed:29599213 / PubMed:29599213 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.8 - 3.0 Å |
| 構造データ | EMDB-7442, PDB-6caj:  EMDB-7443:  EMDB-7444: |
| 化合物 | 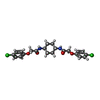 ChemComp-C7B: |
| 由来 |
|
 キーワード キーワード | TRANSLATION / guanine nucleotide exchange factor / translation initiation / ISRIB-bound / decameric complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)