+Search query
-Structure paper
| Title | Conformational basis for substrate recognition and regulation of catalytic activity in Staphylococcus aureus nucleoside di-phosphate kinase. |
|---|---|
| Journal, issue, pages | Biochim. Biophys. Acta, Vol. 1814, Page 1349-1357, Year 2011 |
| Publish date | Jan 6, 2011 (structure data deposition date) |
 Authors Authors | Srivastava, S.K. / Rajasree, K. / Gopal, B. |
 External links External links |  Biochim. Biophys. Acta / Biochim. Biophys. Acta /  PubMed:21745603 PubMed:21745603 |
| Methods | X-ray diffraction |
| Resolution | 2.22 - 2.9 Å |
| Structure data |  PDB-3q83:  PDB-3q86:  PDB-3q89:  PDB-3q8u:  PDB-3q8v:  PDB-3q8y: |
| Chemicals |  ChemComp-HOH:  ChemComp-GTP:  ChemComp-MG: 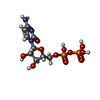 ChemComp-CDP:  ChemComp-ADP:  ChemComp-UDP: 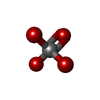 ChemComp-VO4: |
| Source |
|
 Keywords Keywords | TRANSFERASE / Ferridoxin fold / alpha-beta protein family / Nucleoside diphosphate kinases (NDKs) catalyze the transfer of a gamma phosphate from nucleoside triphosphates to nucleoside diphosphate / Nucleotide binding / Metal binding / Phosphorylation / Kinase / Nucleotide metabolism / Nucleoside diphosphate kinases (NDKs) / transfer / a gamma phosphate / nucleoside triphosphates / nucleoside diphosphate / Nucleotide binding Magnesium / Magnesium / gamma phosphate |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




