+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9571 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
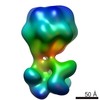
| Title | C. elegans INX-6 gap junction channel | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | innexin / gap junction channel / wild type / TRANSPORT PROTEIN | |||||||||
| Function / homology | gap junction hemi-channel activity / Innexin / Innexin / Pannexin family profile. / gap junction / gap junction channel activity / monoatomic ion transmembrane transport / plasma membrane / Innexin-6 Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Oshima A / Tani K / Fujiyoshi Y | |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2016 Journal: Nat Commun / Year: 2016Title: Atomic structure of the innexin-6 gap junction channel determined by cryo-EM. Authors: Atsunori Oshima / Kazutoshi Tani / Yoshinori Fujiyoshi /  Abstract: Innexins, a large protein family comprising invertebrate gap junction channels, play an essential role in nervous system development and electrical synapse formation. Here we report the cryo-electron ...Innexins, a large protein family comprising invertebrate gap junction channels, play an essential role in nervous system development and electrical synapse formation. Here we report the cryo-electron microscopy structures of Caenorhabditis elegans innexin-6 (INX-6) gap junction channels at atomic resolution. We find that the arrangements of the transmembrane helices and extracellular loops of the INX-6 monomeric structure are highly similar to those of connexin-26 (Cx26), despite the lack of significant sequence similarity. The INX-6 gap junction channel comprises hexadecameric subunits but reveals the N-terminal pore funnel, consistent with Cx26. The helix-rich cytoplasmic loop and C-terminus are intercalated one-by-one through an octameric hemichannel, forming a dome-like entrance that interacts with N-terminal loops in the pore. These observations suggest that the INX-6 cytoplasmic domains are cooperatively associated with the N-terminal funnel conformation, and an essential linkage of the N-terminal with channel activity is presumably preserved across gap junction families. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9571.map.gz emd_9571.map.gz | 28.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9571-v30.xml emd-9571-v30.xml emd-9571.xml emd-9571.xml | 8.7 KB 8.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9571.png emd_9571.png | 263.2 KB | ||
| Filedesc metadata |  emd-9571.cif.gz emd-9571.cif.gz | 4.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9571 http://ftp.pdbj.org/pub/emdb/structures/EMD-9571 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9571 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9571 | HTTPS FTP |
-Validation report
| Summary document |  emd_9571_validation.pdf.gz emd_9571_validation.pdf.gz | 582.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_9571_full_validation.pdf.gz emd_9571_full_validation.pdf.gz | 582 KB | Display | |
| Data in XML |  emd_9571_validation.xml.gz emd_9571_validation.xml.gz | 5.7 KB | Display | |
| Data in CIF |  emd_9571_validation.cif.gz emd_9571_validation.cif.gz | 6.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9571 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9571 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9571 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-9571 | HTTPS FTP |
-Related structure data
| Related structure data |  5h1rMC  9570C  5h1qC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9571.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9571.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.23 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : INX-6
| Entire | Name: INX-6 |
|---|---|
| Components |
|
-Supramolecule #1: INX-6
| Supramolecule | Name: INX-6 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Innexin-6
| Macromolecule | Name: Innexin-6 / type: protein_or_peptide / ID: 1 / Number of copies: 16 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 45.173766 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASQVGAINS VNALISRVFV QPKGDLADRL NSRVTVVILA VSSALLLSSH FIGDPITCWT PAQFNAQWVN FVNQYCFVHG TYFVPLDQQ LAFEEEERTK VSIQYYQWVP YVFALQAFLF YIPRFIWKAM IAYSGYDLAA AVKYVDRFWS ENRDKDDKFK T RLAAFEGR ...String: MASQVGAINS VNALISRVFV QPKGDLADRL NSRVTVVILA VSSALLLSSH FIGDPITCWT PAQFNAQWVN FVNQYCFVHG TYFVPLDQQ LAFEEEERTK VSIQYYQWVP YVFALQAFLF YIPRFIWKAM IAYSGYDLAA AVKYVDRFWS ENRDKDDKFK T RLAAFEGR PSVYIWDGIR LARKKRSRNM ALFYTLSTVW QAVNAWIQFY ILTQLLDSSI YTLWGPSILG DLLQGNDWQT TG HFPRIVH CDFNRRRPAS VQLDTVLCVL TLNIYYEKLF IFLWFWLVFV AVVSTVNCFK WIYYLCNKTK AQKTIKNYLS TAP IKSTIS DDQFFSALGE DGLFIMDQMA LNLGDIPASY LTISMRNICQ DFIESEDYID EERTPFVKSI KHT UniProtKB: Innexin-6 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | JEOL 3000SFF |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 7.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Applied symmetry - Point group: D8 (2x8 fold dihedral) / Resolution.type: BY AUTHOR / Resolution: 3.6 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 1.4) / Number images used: 35608 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















