[English] 日本語
 Yorodumi
Yorodumi- EMDB-50222: Human DNA polymerase epsilon bound to DNA and PCNA (open conformation) -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human DNA polymerase epsilon bound to DNA and PCNA (open conformation) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA / polymerase / epsilon / PCNA / leading strand / human / replication / replisome / proofreading | |||||||||
| Function / homology |  Function and homology information Function and homology informationDNA replication initiation / epsilon DNA polymerase complex / dinucleotide insertion or deletion binding / PCNA-p21 complex / mitotic telomere maintenance via semi-conservative replication / purine-specific mismatch base pair DNA N-glycosylase activity / nucleotide-excision repair, DNA gap filling / nuclear lamina / Polymerase switching / Processive synthesis on the lagging strand ...DNA replication initiation / epsilon DNA polymerase complex / dinucleotide insertion or deletion binding / PCNA-p21 complex / mitotic telomere maintenance via semi-conservative replication / purine-specific mismatch base pair DNA N-glycosylase activity / nucleotide-excision repair, DNA gap filling / nuclear lamina / Polymerase switching / Processive synthesis on the lagging strand / DNA replication proofreading / MutLalpha complex binding / PCNA complex / single-stranded DNA 3'-5' DNA exonuclease activity / Telomere C-strand (Lagging Strand) Synthesis / Removal of the Flap Intermediate / Mismatch repair (MMR) directed by MSH2:MSH3 (MutSbeta) / Mismatch repair (MMR) directed by MSH2:MSH6 (MutSalpha) / Transcription of E2F targets under negative control by DREAM complex / Polymerase switching on the C-strand of the telomere / replisome / Processive synthesis on the C-strand of the telomere / response to L-glutamate / Removal of the Flap Intermediate from the C-strand / Hydrolases; Acting on ester bonds; Exodeoxyribonucleases producing 5'-phosphomonoesters / response to dexamethasone / DNA polymerase processivity factor activity / histone acetyltransferase binding / leading strand elongation / G1/S-Specific Transcription / DNA synthesis involved in DNA repair / nuclear replication fork / replication fork processing / SUMOylation of DNA replication proteins / PCNA-Dependent Long Patch Base Excision Repair / Activation of the pre-replicative complex / embryonic organ development / response to cadmium ion / estrous cycle / mismatch repair / cyclin-dependent protein kinase holoenzyme complex / translesion synthesis / base-excision repair, gap-filling / DNA polymerase binding / epithelial cell differentiation / liver regeneration / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / positive regulation of DNA replication / Translesion synthesis by REV1 / nuclear estrogen receptor binding / positive regulation of DNA repair / Translesion synthesis by POLK / replication fork / Translesion synthesis by POLI / Gap-filling DNA repair synthesis and ligation in GG-NER / male germ cell nucleus / Termination of translesion DNA synthesis / G1/S transition of mitotic cell cycle / Translesion Synthesis by POLH / Recognition of DNA damage by PCNA-containing replication complex / receptor tyrosine kinase binding / DNA-templated DNA replication / HDR through Homologous Recombination (HRR) / cellular response to xenobiotic stimulus / Dual Incision in GG-NER / cellular response to hydrogen peroxide / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / cellular response to UV / response to estradiol / mitotic cell cycle / E3 ubiquitin ligases ubiquitinate target proteins / heart development / 4 iron, 4 sulfur cluster binding / chromatin organization / DNA-directed DNA polymerase / damaged DNA binding / DNA-directed DNA polymerase activity / DNA replication / chromosome, telomeric region / nuclear body / nucleotide binding / chromatin binding / centrosome / chromatin / protein-containing complex binding / enzyme binding / negative regulation of transcription by RNA polymerase II / DNA binding / extracellular exosome / zinc ion binding / nucleoplasm / identical protein binding / nucleus / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
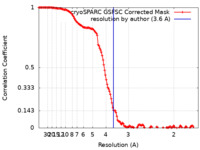
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Roske JJ / Yeeles JTP | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Structural basis for processive daughter-strand synthesis and proofreading by the human leading-strand DNA polymerase Pol ε. Authors: Johann J Roske / Joseph T P Yeeles /  Abstract: During chromosome replication, the nascent leading strand is synthesized by DNA polymerase epsilon (Pol ε), which associates with the sliding clamp processivity factor proliferating cell nuclear ...During chromosome replication, the nascent leading strand is synthesized by DNA polymerase epsilon (Pol ε), which associates with the sliding clamp processivity factor proliferating cell nuclear antigen (PCNA) to form a processive holoenzyme. For high-fidelity DNA synthesis, Pol ε relies on nucleotide selectivity and its proofreading ability to detect and excise a misincorporated nucleotide. Here, we present cryo-electron microscopy (cryo-EM) structures of human Pol ε in complex with PCNA, DNA and an incoming nucleotide, revealing how Pol ε associates with PCNA through its PCNA-interacting peptide box and additional unique features of its catalytic domain. Furthermore, by solving a series of cryo-EM structures of Pol ε at a mismatch-containing DNA, we elucidate how Pol ε senses and edits a misincorporated nucleotide. Our structures delineate steps along an intramolecular switching mechanism between polymerase and exonuclease activities, providing the basis for a proofreading mechanism in B-family replicative polymerases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_50222.map.gz emd_50222.map.gz | 223.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-50222-v30.xml emd-50222-v30.xml emd-50222.xml emd-50222.xml | 22 KB 22 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_50222_fsc.xml emd_50222_fsc.xml | 14.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_50222.png emd_50222.png | 131.6 KB | ||
| Masks |  emd_50222_msk_1.map emd_50222_msk_1.map emd_50222_msk_2.map emd_50222_msk_2.map | 236.9 MB 236.9 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-50222.cif.gz emd-50222.cif.gz | 7 KB | ||
| Others |  emd_50222_additional_1.map.gz emd_50222_additional_1.map.gz emd_50222_half_map_1.map.gz emd_50222_half_map_1.map.gz emd_50222_half_map_2.map.gz emd_50222_half_map_2.map.gz | 223.6 MB 219.8 MB 219.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-50222 http://ftp.pdbj.org/pub/emdb/structures/EMD-50222 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-50222 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-50222 | HTTPS FTP |
-Related structure data
| Related structure data |  9f6dMC  9f6eC  9f6fC  9f6iC  9f6jC  9f6kC  9f6lC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_50222.map.gz / Format: CCP4 / Size: 236.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_50222.map.gz / Format: CCP4 / Size: 236.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.84065 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_50222_msk_1.map emd_50222_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Mask #2
| File |  emd_50222_msk_2.map emd_50222_msk_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Map from focussed refinement with Mask around Polymerase...
| File | emd_50222_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map from focussed refinement with Mask around Polymerase epsilon catalytic domain | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_50222_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_50222_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Quaternary Complex of human leading strand polymerase epsilon, Pr...
+Supramolecule #1: Quaternary Complex of human leading strand polymerase epsilon, Pr...
+Supramolecule #2: DNA polymerase epsilon catalytic subunit A
+Supramolecule #3: Proliferating cell nuclear antigen
+Supramolecule #4: DNA
+Macromolecule #1: DNA polymerase epsilon catalytic subunit A
+Macromolecule #2: Proliferating cell nuclear antigen
+Macromolecule #3: DNA nascent strand
+Macromolecule #4: DNA template strand
+Macromolecule #5: IRON/SULFUR CLUSTER
+Macromolecule #6: 2',3'-dideoxyadenosine triphosphate
+Macromolecule #7: MAGNESIUM ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 40.08 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)


















































 Trichoplusia ni (cabbage looper)
Trichoplusia ni (cabbage looper)