[English] 日本語
 Yorodumi
Yorodumi- EMDB-4339: Electron cryo-microscopy structure of the canonical TRPC4 ion channel -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4339 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Electron cryo-microscopy structure of the canonical TRPC4 ion channel | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ION CHANNEL / TRPC4 / ANKYRIN REPEATS / CIRB DOMAIN / MEMBRANE PROTEIN / TRANSPORT PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationstore-operated calcium channel activity / cation channel complex / inositol 1,4,5 trisphosphate binding / monoatomic cation transport / regulation of cytosolic calcium ion concentration / monoatomic cation channel activity / calcium ion transmembrane transport / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Vinayagam D / Mager T | |||||||||
| Funding support |  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2018 Journal: Elife / Year: 2018Title: Electron cryo-microscopy structure of the canonical TRPC4 ion channel. Authors: Deivanayagabarathy Vinayagam / Thomas Mager / Amir Apelbaum / Arne Bothe / Felipe Merino / Oliver Hofnagel / Christos Gatsogiannis / Stefan Raunser /  Abstract: Canonical transient receptor channels (TRPC) are non-selective cation channels. They are involved in receptor-operated Ca signaling and have been proposed to act as store-operated channels (SOC). ...Canonical transient receptor channels (TRPC) are non-selective cation channels. They are involved in receptor-operated Ca signaling and have been proposed to act as store-operated channels (SOC). Their malfunction is related to cardiomyopathies and their modulation by small molecules has been shown to be effective against renal cancer cells. The molecular mechanism underlying the complex activation and regulation is poorly understood. Here, we report the electron cryo-microscopy structure of zebrafish TRPC4 in its unliganded (apo), closed state at an overall resolution of 3.6 Å. The structure reveals the molecular architecture of the cation conducting pore, including the selectivity filter and lower gate. The cytoplasmic domain contains two key hubs that have been shown to interact with modulating proteins. Structural comparisons with other TRP channels give novel insights into the general architecture and domain organization of this superfamily of channels and help to understand their function and pharmacology. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4339.map.gz emd_4339.map.gz | 4.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4339-v30.xml emd-4339-v30.xml emd-4339.xml emd-4339.xml | 15.7 KB 15.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_4339.png emd_4339.png | 90.5 KB | ||
| Filedesc metadata |  emd-4339.cif.gz emd-4339.cif.gz | 6.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4339 http://ftp.pdbj.org/pub/emdb/structures/EMD-4339 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4339 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4339 | HTTPS FTP |
-Validation report
| Summary document |  emd_4339_validation.pdf.gz emd_4339_validation.pdf.gz | 407.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_4339_full_validation.pdf.gz emd_4339_full_validation.pdf.gz | 407 KB | Display | |
| Data in XML |  emd_4339_validation.xml.gz emd_4339_validation.xml.gz | 5.9 KB | Display | |
| Data in CIF |  emd_4339_validation.cif.gz emd_4339_validation.cif.gz | 6.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4339 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4339 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4339 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4339 | HTTPS FTP |
-Related structure data
| Related structure data |  6g1kMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4339.map.gz / Format: CCP4 / Size: 42.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4339.map.gz / Format: CCP4 / Size: 42.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : TRPC4
| Entire | Name: TRPC4 |
|---|---|
| Components |
|
-Supramolecule #1: TRPC4
| Supramolecule | Name: TRPC4 / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 / Details: Membrane protein |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 432 KDa |
-Macromolecule #1: Transient receptor potential cation channel subfamily c member 4a
| Macromolecule | Name: Transient receptor potential cation channel subfamily c member 4a type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 105.9345 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: GSQLYFRRTD NSSYRDRIPL RIVRAESELS TQEKSYLSAV EKGDYASVKL ALEEAEIYFK ININCIDPLG RTALLIAIEN ENLEIIELL LSFNVYVGDA LLHAIRKEVV GAVELLLNHK KPSGEKQVPP ILLDKQFSDF TPDITPIILA AHTNNYEIIK M LVQKGVSV ...String: GSQLYFRRTD NSSYRDRIPL RIVRAESELS TQEKSYLSAV EKGDYASVKL ALEEAEIYFK ININCIDPLG RTALLIAIEN ENLEIIELL LSFNVYVGDA LLHAIRKEVV GAVELLLNHK KPSGEKQVPP ILLDKQFSDF TPDITPIILA AHTNNYEIIK M LVQKGVSV PQPHEVRCNC VECVSSSDVD SLRHSRSRLN IYKALASPSL IALSSEDPFL TAFQLSWELQ ELSKVENEFK AE YEELSHQ CKHFAKDLLD QTRSSRELEL ILNFRDDMNL LQDEANNELA RLKLAIKYRQ KEFVAQPNCQ QLLASRWYDE FPG WRRRHW AGKLITCVFI GLMFPLLSLC YLVAPKSRYG LFIRKPFIKF ICHTASYLTF LFLLLLASQH IVSNNPDRQG PKPT TVEWM ILPWVLGFIW TEIKQMWDGG FQDYIHDWWN LMDFVMNSLY LATISLKIVA YVKYSGCKPR DTWEMWHPTL VAEAV FAIA NIFSSLRLIS LFTANSHLGP LQISLGRMLL DILKFLFIYC LVLLAFANGL NQLYFYYENS EGMTCKGIRC ERQNNA FST LFETLQSLFW SIFGLISLYV TNVKADHKFT EFVGATMFGT YNVISLVVLL NMLIAMMNNS YQHIADHADI EWKFART KL WMSYFEEGGT LPPPFNIIPS PKSICYLITW IKVHVFKRRS KRTETFGTLG RRAAENVRLN HQYQEVLRNL VKRYVAAM I RDAKTEEGLT EENFKELKQD ISSFRYEVIG MMKGNRKSTR ANKSDTSASD VSHPEGSLQY SSALKQNSKL HLYDVTTAL QQQNSEEAKA SLGCLANGSA VVLTEPILKD KARSDFPKDF TDFGLFPKKQ NPNKIYSLAE EATESDPDIL DWGKEDKPLA GKVEQDVNE SKCLMEEDER VLEEQEMEHI ASSHEHLEVL FQ UniProtKB: Short transient receptor potential channel 4b |
-Macromolecule #2: CHOLESTEROL HEMISUCCINATE
| Macromolecule | Name: CHOLESTEROL HEMISUCCINATE / type: ligand / ID: 2 / Number of copies: 4 / Formula: Y01 |
|---|---|
| Molecular weight | Theoretical: 486.726 Da |
| Chemical component information |  ChemComp-Y01: |
-Macromolecule #3: (2R)-3-(phosphonooxy)propane-1,2-diyl dihexanoate
| Macromolecule | Name: (2R)-3-(phosphonooxy)propane-1,2-diyl dihexanoate / type: ligand / ID: 3 / Number of copies: 4 / Formula: 44E |
|---|---|
| Molecular weight | Theoretical: 368.36 Da |
| Chemical component information | 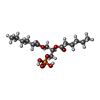 ChemComp-44E: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.25 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Component - Name: PBS |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK III |
| Details | The sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Spherical aberration corrector: CS corrected Microscope |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 4 / Number real images: 3890 / Average exposure time: 12.0 sec. / Average electron dose: 70.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.931 µm / Calibrated defocus min: 0.844 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 1.2 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: OTHER / Overall B value: 90.04 / Target criteria: Real-space correlation | ||||||
| Output model |  PDB-6g1k: |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)