[English] 日本語
 Yorodumi
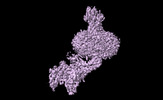
Yorodumi- EMDB-36607: Cryo-EM structure of the glucagon receptor bound to glucagon and ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the glucagon receptor bound to glucagon and beta-arrestin 1 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Complex structure /  glucagon receptor / beta-arrestin 1 / glucagon receptor / beta-arrestin 1 /  glucagon / glucagon /  MEMBRANE PROTEIN MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology information protein C (activated) / renal water retention / Defective AVP does not bind AVPR2 and causes neurohypophyseal diabetes insipidus (NDI) / positive regulation of establishment of endothelial barrier / Vasopressin-like receptors / regulation of systemic arterial blood pressure by vasopressin / protein C (activated) / renal water retention / Defective AVP does not bind AVPR2 and causes neurohypophyseal diabetes insipidus (NDI) / positive regulation of establishment of endothelial barrier / Vasopressin-like receptors / regulation of systemic arterial blood pressure by vasopressin /  vasopressin receptor activity / vasopressin receptor activity /  glucagon receptor binding / negative regulation of coagulation / regulation of glycogen metabolic process ... glucagon receptor binding / negative regulation of coagulation / regulation of glycogen metabolic process ... protein C (activated) / renal water retention / Defective AVP does not bind AVPR2 and causes neurohypophyseal diabetes insipidus (NDI) / positive regulation of establishment of endothelial barrier / Vasopressin-like receptors / regulation of systemic arterial blood pressure by vasopressin / protein C (activated) / renal water retention / Defective AVP does not bind AVPR2 and causes neurohypophyseal diabetes insipidus (NDI) / positive regulation of establishment of endothelial barrier / Vasopressin-like receptors / regulation of systemic arterial blood pressure by vasopressin /  vasopressin receptor activity / vasopressin receptor activity /  glucagon receptor binding / negative regulation of coagulation / regulation of glycogen metabolic process / glucagon receptor binding / negative regulation of coagulation / regulation of glycogen metabolic process /  glucagon receptor activity / positive regulation of systemic arterial blood pressure / glucagon receptor activity / positive regulation of systemic arterial blood pressure /  hemostasis / telencephalon development / negative regulation of execution phase of apoptosis / negative regulation of blood coagulation / hemostasis / telencephalon development / negative regulation of execution phase of apoptosis / negative regulation of blood coagulation /  feeding behavior / feeding behavior /  : / positive regulation of intracellular signal transduction / response to starvation / cellular response to glucagon stimulus / positive regulation of calcium ion import / : / positive regulation of intracellular signal transduction / response to starvation / cellular response to glucagon stimulus / positive regulation of calcium ion import /  exocytosis / positive regulation of insulin secretion involved in cellular response to glucose stimulus / exocytosis / positive regulation of insulin secretion involved in cellular response to glucose stimulus /  peptide hormone binding / regulation of insulin secretion / endocytic vesicle / Synthesis, secretion, and deacylation of Ghrelin / Gamma-carboxylation of protein precursors / Transport of gamma-carboxylated protein precursors from the endoplasmic reticulum to the Golgi apparatus / Common Pathway of Fibrin Clot Formation / Removal of aminoterminal propeptides from gamma-carboxylated proteins / protein kinase A signaling / positive regulation of vasoconstriction / positive regulation of gluconeogenesis / cellular response to hormone stimulus / activation of adenylate cyclase activity / Intrinsic Pathway of Fibrin Clot Formation / cellular response to starvation / hormone-mediated signaling pathway / response to nutrient / guanyl-nucleotide exchange factor activity / peptide hormone binding / regulation of insulin secretion / endocytic vesicle / Synthesis, secretion, and deacylation of Ghrelin / Gamma-carboxylation of protein precursors / Transport of gamma-carboxylated protein precursors from the endoplasmic reticulum to the Golgi apparatus / Common Pathway of Fibrin Clot Formation / Removal of aminoterminal propeptides from gamma-carboxylated proteins / protein kinase A signaling / positive regulation of vasoconstriction / positive regulation of gluconeogenesis / cellular response to hormone stimulus / activation of adenylate cyclase activity / Intrinsic Pathway of Fibrin Clot Formation / cellular response to starvation / hormone-mediated signaling pathway / response to nutrient / guanyl-nucleotide exchange factor activity /  viral budding from plasma membrane / response to activity / positive regulation of peptidyl-threonine phosphorylation / generation of precursor metabolites and energy / response to cytokine / viral budding from plasma membrane / response to activity / positive regulation of peptidyl-threonine phosphorylation / generation of precursor metabolites and energy / response to cytokine /  gluconeogenesis / Cell surface interactions at the vascular wall / gluconeogenesis / Cell surface interactions at the vascular wall /  peptide binding / peptide binding /  Post-translational protein phosphorylation / clathrin-coated endocytic vesicle membrane / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Glucagon signaling in metabolic regulation / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Post-translational protein phosphorylation / clathrin-coated endocytic vesicle membrane / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Glucagon signaling in metabolic regulation / adenylate cyclase-activating G protein-coupled receptor signaling pathway /  hormone activity / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins / hormone activity / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Glucagon-type ligand receptors / Vasopressin regulates renal water homeostasis via Aquaporins /  regulation of blood pressure / negative regulation of inflammatory response / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Golgi lumen / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / regulation of blood pressure / negative regulation of inflammatory response / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Golgi lumen / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) /  blood coagulation / Cargo recognition for clathrin-mediated endocytosis / blood coagulation / Cargo recognition for clathrin-mediated endocytosis /  glucose homeostasis / positive regulation of peptidyl-serine phosphorylation / glucose homeostasis / positive regulation of peptidyl-serine phosphorylation /  Clathrin-mediated endocytosis / G alpha (s) signalling events / G alpha (q) signalling events / clathrin-dependent endocytosis of virus by host cell / secretory granule lumen / cell surface receptor signaling pathway / positive regulation of ERK1 and ERK2 cascade / host cell surface receptor binding / Clathrin-mediated endocytosis / G alpha (s) signalling events / G alpha (q) signalling events / clathrin-dependent endocytosis of virus by host cell / secretory granule lumen / cell surface receptor signaling pathway / positive regulation of ERK1 and ERK2 cascade / host cell surface receptor binding /  endosome / G protein-coupled receptor signaling pathway / fusion of virus membrane with host plasma membrane / negative regulation of cell population proliferation / endosome / G protein-coupled receptor signaling pathway / fusion of virus membrane with host plasma membrane / negative regulation of cell population proliferation /  endoplasmic reticulum lumen / endoplasmic reticulum lumen /  signaling receptor binding / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane / signaling receptor binding / serine-type endopeptidase activity / fusion of virus membrane with host endosome membrane /  viral envelope / viral envelope /  calcium ion binding / positive regulation of cell population proliferation / virion attachment to host cell / positive regulation of gene expression / negative regulation of apoptotic process / host cell plasma membrane / perinuclear region of cytoplasm / virion membrane / calcium ion binding / positive regulation of cell population proliferation / virion attachment to host cell / positive regulation of gene expression / negative regulation of apoptotic process / host cell plasma membrane / perinuclear region of cytoplasm / virion membrane /  Golgi apparatus / Golgi apparatus /  endoplasmic reticulum / endoplasmic reticulum /  proteolysis / proteolysis /  extracellular space / extracellular region / extracellular space / extracellular region /  membrane / identical protein binding membrane / identical protein bindingSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.3 Å cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Chen K / Zhang C / Lin S / Zhao Q / Wu B | |||||||||
| Funding support |  China, 2 items China, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: Tail engagement of arrestin at the glucagon receptor. Authors: Kun Chen / Chenhui Zhang / Shuling Lin / Xinyu Yan / Heng Cai / Cuiying Yi / Limin Ma / Xiaojing Chu / Yuchen Liu / Ya Zhu / Shuo Han / Qiang Zhao / Beili Wu /  Abstract: Arrestins have pivotal roles in regulating G protein-coupled receptor (GPCR) signalling by desensitizing G protein activation and mediating receptor internalization. It has been proposed that the ...Arrestins have pivotal roles in regulating G protein-coupled receptor (GPCR) signalling by desensitizing G protein activation and mediating receptor internalization. It has been proposed that the arrestin binds to the receptor in two different conformations, 'tail' and 'core', which were suggested to govern distinct processes of receptor signalling and trafficking. However, little structural information is available for the tail engagement of the arrestins. Here we report two structures of the glucagon receptor (GCGR) bound to β-arrestin 1 (βarr1) in glucagon-bound and ligand-free states. These structures reveal a receptor tail-engaged binding mode of βarr1 with many unique features, to our knowledge, not previously observed. Helix VIII, instead of the receptor core, has a major role in accommodating βarr1 by forming extensive interactions with the central crest of βarr1. The tail-binding pose is further defined by a close proximity between the βarr1 C-edge and the receptor helical bundle, and stabilized by a phosphoinositide derivative that bridges βarr1 with helices I and VIII of GCGR. Lacking any contact with the arrestin, the receptor core is in an inactive state and loosely binds to glucagon. Further functional studies suggest that the tail conformation of GCGR-βarr governs βarr recruitment at the plasma membrane and endocytosis of GCGR, and provides a molecular basis for the receptor forming a super-complex simultaneously with G protein and βarr to promote sustained signalling within endosomes. These findings extend our knowledge about the arrestin-mediated modulation of GPCR functionalities. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36607.map.gz emd_36607.map.gz | 57.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36607-v30.xml emd-36607-v30.xml emd-36607.xml emd-36607.xml | 18.4 KB 18.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36607.png emd_36607.png | 46.9 KB | ||
| Others |  emd_36607_half_map_1.map.gz emd_36607_half_map_1.map.gz emd_36607_half_map_2.map.gz emd_36607_half_map_2.map.gz | 59.4 MB 59.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36607 http://ftp.pdbj.org/pub/emdb/structures/EMD-36607 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36607 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36607 | HTTPS FTP |
-Related structure data
| Related structure data |  8jrvMC  8jruC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36607.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36607.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.071 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_36607_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
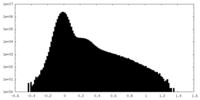
| Projections & Slices |
| ||||||||||||
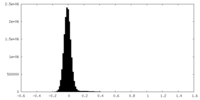
| Density Histograms |
-Half map: #1
| File | emd_36607_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
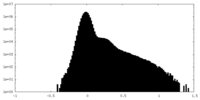
| Projections & Slices |
| ||||||||||||
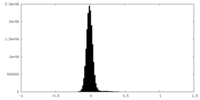
| Density Histograms |
- Sample components
Sample components
-Entire : The glucagon receptor bound to glucagon and beta-arrestin 1
| Entire | Name: The glucagon receptor bound to glucagon and beta-arrestin 1 |
|---|---|
| Components |
|
-Supramolecule #1: The glucagon receptor bound to glucagon and beta-arrestin 1
| Supramolecule | Name: The glucagon receptor bound to glucagon and beta-arrestin 1 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: HA signal peptide,HPC4 purification tag,Glucagon receptor,C-termi...
| Macromolecule | Name: HA signal peptide,HPC4 purification tag,Glucagon receptor,C-terminal tail of Vasopressin V2 receptor type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 54.163012 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MKTIIALSYI FCLVFAGAPE DQVDPRLIDG KGSGSAGSAG SQVMDFLFEK WKLYGDQCHH NLSLLPPPTE LVCNRTFDKY SCWPDTPAN TTANISCPWY LPWHHKVQHR FVFKRCGPDG QWVRGPRGQP WRDASQCQMD GEEIEVQKEV AKMYSSFQVM Y TVGYSLSL ...String: MKTIIALSYI FCLVFAGAPE DQVDPRLIDG KGSGSAGSAG SQVMDFLFEK WKLYGDQCHH NLSLLPPPTE LVCNRTFDKY SCWPDTPAN TTANISCPWY LPWHHKVQHR FVFKRCGPDG QWVRGPRGQP WRDASQCQMD GEEIEVQKEV AKMYSSFQVM Y TVGYSLSL GALLLALAIL GGLSKLHCTR NAIHANLFAS FVLKASSVLV IDGLLRTRYS QKIGDDLSVS TWLSDGAVAG CR VAAVFMQ YGIVANYCWL LVEGLYLHNL LGLATLPERS FFSLYLGIGW GAPMLFVVPW AVVKCLFENV QCWTSNDNMG FWW ILRFPV FLAILINFFI FVRIVQLLVA KLRARQMHHT DYKFRLAKST LTLIPLLGVH EVVFAFVTDE HAQGTLRSAK LFFD LFLSS FQGLLVAVLY CFLNKEVQSE LRRRWHRWRL GKVLWEERNT SNARGRTPPS LGPQDE(SEP)CT(TPO) A(SEP) (SEP)(SEP)LAKDT SS UniProtKB:  Hemagglutinin, Vitamin K-dependent protein C, Hemagglutinin, Vitamin K-dependent protein C,  Glucagon receptor, Glucagon receptor,  Vasopressin V2 receptor Vasopressin V2 receptor |
-Macromolecule #2: Glucagon
| Macromolecule | Name: Glucagon / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 3.486781 KDa |
| Sequence | String: HSQGTFTSDY SKYLDSRRAQ DFVQWLMNT UniProtKB: Pro-glucagon |
-Macromolecule #3: Beta-arrestin 1 and single-chain fragment variable 30 (scFv30)
| Macromolecule | Name: Beta-arrestin 1 and single-chain fragment variable 30 (scFv30) type: protein_or_peptide / ID: 3 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 69.173891 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVEPVDGV VLVDPEYLKE RRVYVTLTAA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPSSVTL QPGPEDTGKA IGVDYEVKAF VAENLEEKIH K RNSVRLVI ...String: MGDKGTRVFK KASPNGKLTV YLGKRDFVDH IDLVEPVDGV VLVDPEYLKE RRVYVTLTAA FRYGREDLDV LGLTFRKDLF VANVQSFPP APEDKKPLTR LQERLIKKLG EHAYPFTFEI PPNLPSSVTL QPGPEDTGKA IGVDYEVKAF VAENLEEKIH K RNSVRLVI EKVQYAPERP GPQPTAETTR QFLMSDKPLH LEASLDKEIY YHGEPISVNV HVTNNTNKTV KKIKISVRQY AD IVLFNTA QYKVPVAMEE ADDTVAPSST FSKVYTLTPF LANNREKRGL ALDGKLKHED TNLASSTLLR EGANREILGI IVS YKVKVK LVVSRGGLLG DLASSDVAVE LPFTLMHPKP KEEPPHREVP EHETPVDTNL SDIQMTQSPS SLSASVGDRV TITC RASQS VSSAVAWYQQ KPGKAPKLLI YSASSLYSGV PSRFSGSRSG TDFTLTISSL QPEDFATYYC QQYKYVPVTF GQGTK VEIK GTTAASGSSG GSSSGAEVQL VESGGGLVQP GGSLRLSCAA SGFNVYSSSI HWVRQAPGKG LEWVASISSY YGYTYY ADS VKGRFTISAD TSKNTAYLQM NSLRAEDTAV YYCARSRQFW YSGLDYWGQG TLVTVSSAHH HHHH |
-Macromolecule #4: Nanobody 32
| Macromolecule | Name: Nanobody 32 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Molecular weight | Theoretical: 13.867408 KDa |
| Recombinant expression | Organism:   Escherichia coli BL21 (bacteria) Escherichia coli BL21 (bacteria) |
| Sequence | String: MAQVQLQESG GGLVQAGGSL RLSCVVSGFF FDTVTMAWYR RAPGKHRELV ASATAGGTTT YADSVKDRFT ISRDNAKNTV YLQMNSLKP EDTAVYYCNT FVRSLSWGQG TQVTVSSHHH HHHEPEA |
-Macromolecule #5: [(2R)-2-octanoyloxy-3-[oxidanyl-[(1R,2R,3S,4R,5R,6S)-2,3,6-tris(o...
| Macromolecule | Name: [(2R)-2-octanoyloxy-3-[oxidanyl-[(1R,2R,3S,4R,5R,6S)-2,3,6-tris(oxidanyl)-4,5-diphosphonooxy-cyclohexyl]oxy-phosphoryl]oxy-propyl] octanoate type: ligand / ID: 5 / Number of copies: 1 / Formula: PIO |
|---|---|
| Molecular weight | Theoretical: 746.566 Da |
| Chemical component information |  ChemComp-PIO: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 6 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.8 µm Bright-field microscopy / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.8 µm |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 70.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 300738 |
 Movie
Movie Controller
Controller


































 Z
Z Y
Y X
X