[English] 日本語
 Yorodumi
Yorodumi- EMDB-33592: Cryo-EM structure of Arabidopsis thaliana SOS1 in an occluded state -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of Arabidopsis thaliana SOS1 in an occluded state | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Sodium/proton antiporter / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationpotassium:proton antiporter activity / sodium:proton antiporter activity / chloroplast envelope / sodium ion import across plasma membrane / regulation of reactive oxygen species metabolic process / sodium ion transport / response to salt stress / potassium ion transmembrane transport / regulation of intracellular pH / response to reactive oxygen species ...potassium:proton antiporter activity / sodium:proton antiporter activity / chloroplast envelope / sodium ion import across plasma membrane / regulation of reactive oxygen species metabolic process / sodium ion transport / response to salt stress / potassium ion transmembrane transport / regulation of intracellular pH / response to reactive oxygen species / response to hydrogen peroxide / response to oxidative stress / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||
 Authors Authors | Wang Y / Zhao Y / Gao Y | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Architecture and autoinhibitory mechanism of the plasma membrane Na/H antiporter SOS1 in Arabidopsis. Authors: Yuhang Wang / Chengcai Pan / Qihao Chen / Qing Xie / Yiwei Gao / Lingli He / Yue Li / Yanli Dong / Xingyu Jiang / Yan Zhao /  Abstract: Salt-overly-sensitive 1 (SOS1) is a unique electroneutral Na/H antiporter at the plasma membrane of higher plants and plays a central role in resisting salt stress. SOS1 is kept in a resting state ...Salt-overly-sensitive 1 (SOS1) is a unique electroneutral Na/H antiporter at the plasma membrane of higher plants and plays a central role in resisting salt stress. SOS1 is kept in a resting state with basal activity and activated upon phosphorylation. Here, we report the structures of SOS1. SOS1 forms a homodimer, with each monomer composed of transmembrane and intracellular domains. We find that SOS1 is locked in an occluded state by shifting of the lateral-gate TM5b toward the dimerization domain, thus shielding the Na/H binding site. We speculate that the dimerization of the intracellular domain is crucial to stabilize the transporter in this specific conformation. Moreover, two discrete fragments and a residue W1013 are important to prevent the transition of SOS1 to an alternative conformational state, as validated by functional complementation assays. Our study enriches understanding of the alternate access model of eukaryotic Na/H exchangers. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33592.map.gz emd_33592.map.gz | 118.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33592-v30.xml emd-33592-v30.xml emd-33592.xml emd-33592.xml | 17 KB 17 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33592.png emd_33592.png | 41.3 KB | ||
| Filedesc metadata |  emd-33592.cif.gz emd-33592.cif.gz | 6.3 KB | ||
| Others |  emd_33592_half_map_1.map.gz emd_33592_half_map_1.map.gz emd_33592_half_map_2.map.gz emd_33592_half_map_2.map.gz | 115.8 MB 115.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33592 http://ftp.pdbj.org/pub/emdb/structures/EMD-33592 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33592 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33592 | HTTPS FTP |
-Related structure data
| Related structure data |  7y3eMC  8hyaC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33592.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33592.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.04 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_33592_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
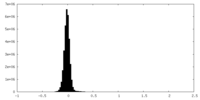
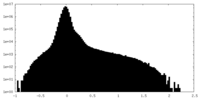
| Density Histograms |
-Half map: #2
| File | emd_33592_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
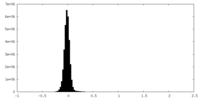
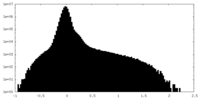
| Density Histograms |
- Sample components
Sample components
-Entire : Arabidopsis sodium/hydrogen exchanger 7 (SOS1), homodimer
| Entire | Name: Arabidopsis sodium/hydrogen exchanger 7 (SOS1), homodimer |
|---|---|
| Components |
|
-Supramolecule #1: Arabidopsis sodium/hydrogen exchanger 7 (SOS1), homodimer
| Supramolecule | Name: Arabidopsis sodium/hydrogen exchanger 7 (SOS1), homodimer type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Sodium/hydrogen exchanger 7
| Macromolecule | Name: Sodium/hydrogen exchanger 7 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 127.327891 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MTTVIDATMA YRFLEEATDS SSSSSSSKLE SSPVDAVLFV GMSLVLGIAS RHLLRGTRVP YTVALLVIGI ALGSLEYGAK HNLGKIGHG IRIWNEIDPE LLLAVFLPAL LFESSFSMEV HQIKRCLGQM VLLAVPGVLI STACLGSLVK VTFPYEWDWK T SLLLGGLL ...String: MTTVIDATMA YRFLEEATDS SSSSSSSKLE SSPVDAVLFV GMSLVLGIAS RHLLRGTRVP YTVALLVIGI ALGSLEYGAK HNLGKIGHG IRIWNEIDPE LLLAVFLPAL LFESSFSMEV HQIKRCLGQM VLLAVPGVLI STACLGSLVK VTFPYEWDWK T SLLLGGLL SATDPVAVVA LLKELGASKK LSTIIEGESL MNDGTAIVVF QLFLKMAMGQ NSDWSSIIKF LLKVALGAVG IG LAFGIAS VIWLKFIFND TVIEITLTIA VSYFAYYTAQ EWAGASGVLT VMTLGMFYAA FARTAFKGDS QKSLHHFWEM VAY IANTLI FILSGVVIAE GILDSDKIAY QGNSWRFLFL LYVYIQLSRV VVVGVLYPLL CRFGYGLDWK ESIILVWSGL RGAV ALALS LSVKQSSGNS HISKETGTLF LFFTGGIVFL TLIVNGSTTQ FVLRLLRMDI LPAPKKRILE YTKYEMLNKA LRAFQ DLGD DEELGPADWP TVESYISSLK GSEGELVHHP HNGSKIGSLD PKSLKDIRMR FLNGVQATYW EMLDEGRISE VTANIL MQS VDEALDQVST TLCDWRGLKP HVNFPNYYNF LHSKVVPRKL VTYFAVERLE SACYISAAFL RAHTIARQQL YDFLGES NI GSIVINESEK EGEEAKKFLE KVRSSFPQVL RVVKTKQVTY SVLNHLLGYI ENLEKVGLLE EKEIAHLHDA VQTGLKKL L RNPPIVKLPK LSDMITSHPL SVALPPAFCE PLKHSKKEPM KLRGVTLYKE GSKPTGVWLI FDGIVKWKSK ILSNNHSLH PTFSHGSTLG LYEVLTGKPY LCDLITDSMV LCFFIDSEKI LSLQSDSTID DFLWQESALV LLKLLRPQIF ESVAMQELRA LVSTESSKL TTYVTGESIE IDCNSIGLLL EGFVKPVGIK EELISSPAAL SPSNGNQSFH NSSEASGIMR VSFSQQATQY I VETRARAI IFNIGAFGAD RTLHRRPSSL TPPRSSSSDQ LQRSFRKEHR GLMSWPENIY AKQQQEINKT TLSLSERAMQ LS IFGSMVN VYRRSVSFGG IYNNKLQDNL LYKKLPLNPA QGLVSAKSES SIVTKKQLET RKHACQLPLK GESSTRQNTM VES SDEEDE DEGIVVRIDS PSKIVFRNDL UniProtKB: Sodium/hydrogen exchanger 7 |
-Macromolecule #2: HEXADECANE
| Macromolecule | Name: HEXADECANE / type: ligand / ID: 2 / Number of copies: 4 / Formula: R16 |
|---|---|
| Molecular weight | Theoretical: 226.441 Da |
| Chemical component information |  ChemComp-R16: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 22.0 µm / Nominal defocus min: 12.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)