+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | ADGRL3/Gi complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationlocomotion involved in locomotory behavior / Rho-activating G protein-coupled receptor signaling pathway / excitatory synapse assembly / cell adhesion mediator activity / : / maintenance of postsynaptic specialization structure / positive regulation of synapse assembly / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration ...locomotion involved in locomotory behavior / Rho-activating G protein-coupled receptor signaling pathway / excitatory synapse assembly / cell adhesion mediator activity / : / maintenance of postsynaptic specialization structure / positive regulation of synapse assembly / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration / positive regulation of relaxation of smooth muscle / Adenylate cyclase inhibitory pathway / D2 dopamine receptor binding / synapse assembly / adenylate cyclase-inhibiting serotonin receptor signaling pathway / G protein-coupled serotonin receptor binding / cellular response to forskolin / regulation of mitotic spindle organization / chemokine-mediated signaling pathway / response to cocaine / Regulation of insulin secretion / neuropeptide signaling pathway / response to prostaglandin E / molecular condensate scaffold activity / positive regulation of cholesterol biosynthetic process / negative regulation of insulin secretion / synapse organization / G protein-coupled receptor binding / response to peptide hormone / G protein-coupled receptor activity / Schaffer collateral - CA1 synapse / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / neuron migration / G-protein beta/gamma-subunit complex binding / centriolar satellite / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G protein-coupled acetylcholine receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / cell-cell junction / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / GDP binding / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / G-protein beta-subunit binding / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / extracellular vesicle / sensory perception of taste / adenylate cyclase-activating G protein-coupled receptor signaling pathway / sperm principal piece / Thrombin signalling through proteinase activated receptors (PARs) / signaling receptor complex adaptor activity / carbohydrate binding / retina development in camera-type eye / GTPase binding / G protein activity / fibroblast proliferation / Ca2+ pathway / midbody / cell cortex / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / G alpha (i) signalling events / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (q) signalling events / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / Ras protein signal transduction / postsynaptic membrane / cell surface receptor signaling pathway / cell population proliferation / Extra-nuclear estrogen signaling / ciliary basal body / G protein-coupled receptor signaling pathway / cell division Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
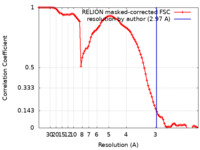
| Method | single particle reconstruction / cryo EM / Resolution: 2.97 Å | |||||||||
 Authors Authors | He Y / Qian Y | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2022 Journal: Mol Cell / Year: 2022Title: Structural insights into adhesion GPCR ADGRL3 activation and G, G, G, and G coupling. Authors: Yu Qian / Zhengxiong Ma / Chunhong Liu / Xinzhi Li / Xinyan Zhu / Na Wang / Zhenmei Xu / Ruixue Xia / Jiale Liang / Yaning Duan / Han Yin / Yangjie Xiong / Anqi Zhang / Changyou Guo / Zheng ...Authors: Yu Qian / Zhengxiong Ma / Chunhong Liu / Xinzhi Li / Xinyan Zhu / Na Wang / Zhenmei Xu / Ruixue Xia / Jiale Liang / Yaning Duan / Han Yin / Yangjie Xiong / Anqi Zhang / Changyou Guo / Zheng Chen / Zhiwei Huang / Yuanzheng He /  Abstract: Adhesion G-protein-coupled receptors (aGPCRs) play key roles in a diversity of physiologies. A hallmark of aGPCR activation is the removal of the inhibitory GAIN domain and the dipping of the cleaved ...Adhesion G-protein-coupled receptors (aGPCRs) play key roles in a diversity of physiologies. A hallmark of aGPCR activation is the removal of the inhibitory GAIN domain and the dipping of the cleaved stalk peptide into the ligand-binding pocket of receptors; however, the detailed mechanism remains obscure. Here, we present cryoelectron microscopy (cryo-EM) structures of ADGRL3 in complex with G, G, G, and G. The structures reveal unique ligand-engaging mode, distinctive activation conformation, and key mechanisms of aGPCR activation. The structures also reveal the uncharted structural information of GPCR/G coupling. A comparison of G, G, G, and G engagements with ADGRL3 reveals the key determinant of G-protein coupling on the far end of αH5 of Gα. A detailed analysis of the engagements allows us to design mutations that specifically enhance one pathway over others. Taken together, our study lays the groundwork for understanding aGPCR activation and G-protein-coupling selectivity. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_32890.map.gz emd_32890.map.gz | 56.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-32890-v30.xml emd-32890-v30.xml emd-32890.xml emd-32890.xml | 18 KB 18 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_32890_fsc.xml emd_32890_fsc.xml | 9.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_32890.png emd_32890.png | 73.3 KB | ||
| Filedesc metadata |  emd-32890.cif.gz emd-32890.cif.gz | 7.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-32890 http://ftp.pdbj.org/pub/emdb/structures/EMD-32890 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32890 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-32890 | HTTPS FTP |
-Related structure data
| Related structure data |  7wybMC  7wy5C  7wy8C  7x10C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_32890.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_32890.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : GPCR/G-protein complex
| Entire | Name: GPCR/G-protein complex |
|---|---|
| Components |
|
-Supramolecule #1: GPCR/G-protein complex
| Supramolecule | Name: GPCR/G-protein complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Supramolecule #2: GPCR/G-protein complex
| Supramolecule | Name: GPCR/G-protein complex / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: ADGRL3
| Supramolecule | Name: ADGRL3 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #5 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Guanine nucleotide-binding protein G(i) subunit alpha-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(i) subunit alpha-1 type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.414047 KDa |
| Recombinant expression | Organism:  Insect BA phytoplasma (bacteria) Insect BA phytoplasma (bacteria) |
| Sequence | String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKNTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKNTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTTGIVETH FTFKDLHFKM FDVGAQRSER KKWIHCFEGV TAIIFCVALS DYDLVLAEDE EM NRMHASM KLFDSICNNK WFTDTSIILF LNKKDLFEEK IKKSPLTICY PEYAGSNTYE EAAAYIQCQF EDLNKRKDTK EIY THFTCS TDTKNVQFVF DAVTDVIIKN NLKDCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.915496 KDa |
| Recombinant expression | Organism:  Insect BA phytoplasma (bacteria) Insect BA phytoplasma (bacteria) |
| Sequence | String: MGSLLQSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD ...String: MGSLLQSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD TTCALWDIET GQQTTTFTGH TGDVMSLSLA PDTRLFVSGA CDASAKLWDV REGMCRQTFT GHESDINAIC FF PNGNAFA TGSDDATCRL FDLRADQELM TYSHDNIICG ITSVSFSKSG RLLLAGYDDF NCNVWDALKA DRAGVLAGHD NRV SCLGVT DDGMAVATGS WDSFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  Insect BA phytoplasma (bacteria) Insect BA phytoplasma (bacteria) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: scFv16
| Macromolecule | Name: scFv16 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 26.277299 KDa |
| Recombinant expression | Organism:  Insect BA phytoplasma (bacteria) Insect BA phytoplasma (bacteria) |
| Sequence | String: VQLVESGGGL VQPGGSRKLS CSASGFAFSS FGMHWVRQAP EKGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSED TAMYYCVRSI YYYGSSPFDF WGQGTTLTVS AGGGGSGGGG SGGGGSADIV MTQATSSVPV TPGESVSISC R SSKSLLHS ...String: VQLVESGGGL VQPGGSRKLS CSASGFAFSS FGMHWVRQAP EKGLEWVAYI SSGSGTIYYA DTVKGRFTIS RDDPKNTLFL QMTSLRSED TAMYYCVRSI YYYGSSPFDF WGQGTTLTVS AGGGGSGGGG SGGGGSADIV MTQATSSVPV TPGESVSISC R SSKSLLHS NGNTYLYWFL QRPGQSPQLL IYRMSNLASG VPDRFSGSGS GTAFTLTISR LEAEDVGVYY CMQHLEYPLT FG AGTKLEL |
-Macromolecule #5: Isoform 3 of Adhesion G protein-coupled receptor L3
| Macromolecule | Name: Isoform 3 of Adhesion G protein-coupled receptor L3 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 172.044625 KDa |
| Recombinant expression | Organism:  Insect BA phytoplasma (bacteria) Insect BA phytoplasma (bacteria) |
| Sequence | String: MWPPQLLILT MLLAPVVHGG KHNERHPALA APLRHAERSP GGALPPRHLL QQPAAERSTA HRGQGPRGAA RGVRGPGAPG AQIAAQAFS RAPIPMAVVR RELSCESYPI ELRCPGTDVI MIESANYGRT DDKICDSDPA QMENIRCYLP DAYKIMSQRC N NRTQCAVV ...String: MWPPQLLILT MLLAPVVHGG KHNERHPALA APLRHAERSP GGALPPRHLL QQPAAERSTA HRGQGPRGAA RGVRGPGAPG AQIAAQAFS RAPIPMAVVR RELSCESYPI ELRCPGTDVI MIESANYGRT DDKICDSDPA QMENIRCYLP DAYKIMSQRC N NRTQCAVV AGPDVFPDPC PGTYKYLEVQ YECVPYKVEQ KVFLCPGLLK GVYQSEHLFE SDHQSGAWCK DPLQASDKIY YM PWTPYRT DTLTEYSSKD DFIAGRPTTT YKLPHRVDGT GFVVYDGALF FNKERTRNIV KFDLRTRIKS GEAIIANANY HDT SPYRWG GKSDIDLAVD ENGLWVIYAT EQNNGKIVIS QLNPYTLRIE GTWDTAYDKR SASNAFMICG ILYVVKSVYE DDDN EATGN KIDYIYNTDQ SKDSLVDVPF PNSYQYIAAV DYNPRDNLLY VWNNYHVVKY SLDFGPLDSR SGPVHHGQVS YISPP IHLD SELERPPVRG ISTTGSLGMG STTTSTTLRT TTWNIGRSTT ASLPGRRNRS TSTPSPAVEV LDDVTTHLPS AASQIP AME ESCEAVEARE IMWFKTRQGQ VAKQPCPAGT IGVSTYLCLA PDGIWDPQGP DLSNCSSPWV NHITQKLKSG ETAANIA RE LAEQTRNHLN AGDITYSVRA MDQLVGLLDV QLRNLTPGGK DSAARSLNKL QKRERSCRAY VQAMVETVNN LLQPQALN A WRDLTTSDQL RAATMLLDTV EESAFVLADN LLKTDIVREN TDNIQLEVAR LSTEGNLEDL KFPENMGHGS TIQLSANTL KQNGRNGEIR VAFVLYNNLG PYLSTENASM KLGTEAMSTN HSVIVNSPVI TAAINKEFSN KVYLADPVVF TVKHIKQSEE NFNPNCSFW SYSKRTMTGY WSTQGCRLLT TNKTHTTCSC NHLTNFAVLM AHVEVKHSDA VHDLLLDVIT WVGILLSLVC L LICIFTFC FFRGLQSDRN TIHKNLCISL FVAELLFLIG INRTDQPIAC AVFAALLHFF FLAAFTWMFL EGVQLYIMLV EV FESEHSR RKYFYLVGYG MPALIVAVSA AVDYRSYGTD KVCWLRLDTY FIWSFIGPAT LIIMLNVIFL GIALYKMFHH TAI LKPESG CLDNINYEDN RPFIKSWVIG AIALLCLLGL TWAFGLMYIN ESTVIMAYLF TIFNSLQGMF IFIFHCVLQK KVRK EYGKC LRTHCCSGKS TESSIGSGKT SGSRTPGRYS TGSQSRIRRM WNDTVRKQSE SSFITGDINS SASLNREPYR ETKGL LNNA RDTSVMDTLP LNGNHGNSYS IAGGEYLSNC VQIIDRGYNH NETALEKKIL KELTSNYIPS YLNNHERSSE QNRNMM NKL VNNLGSGSED DAIVLDDAAS FNHEESLGLE LIHEESDAPL LPPRVYSTDN HQPHHYSRRR FPQDHSESFF PLLTDEH TE DLQSPHRDSL YTSMPALAGV PAADSVTTST QTEAAAAKGG DAEDVYYKSM PNLGSRNHVH PLHAYYQLGR GSSDGFIV P PNKDGASPEG TSKGPAHLVT SL UniProtKB: Adhesion G protein-coupled receptor L3 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | 3D array |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.3000000000000003 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)