[English] 日本語
 Yorodumi
Yorodumi- EMDB-29877: Exploiting Activation and Inactivation Mechanisms in Type I-C CRI... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Exploiting Activation and Inactivation Mechanisms in Type I-C CRISPR-Cas3 for Genome Editing Applications | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CRISPR / type I-C / cascade / anti-CRISPR / HYDROLASE-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationmaintenance of CRISPR repeat elements / endonuclease activity / defense response to virus / Hydrolases; Acting on ester bonds / hydrolase activity / RNA binding Similarity search - Function | |||||||||
| Biological species |  Neisseria lactamica (bacteria) / Neisseria lactamica (bacteria) /  Rhodobacter phage RcNL1 (virus) Rhodobacter phage RcNL1 (virus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Hu C / Nam KH / Ke A | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2024 Journal: Mol Cell / Year: 2024Title: Exploiting activation and inactivation mechanisms in type I-C CRISPR-Cas3 for genome-editing applications. Authors: Chunyi Hu / Mason T Myers / Xufei Zhou / Zhonggang Hou / Macy L Lozen / Ki Hyun Nam / Yan Zhang / Ailong Ke /    Abstract: Type I CRISPR-Cas systems utilize the RNA-guided Cascade complex to identify matching DNA targets and the nuclease-helicase Cas3 to degrade them. Among the seven subtypes, type I-C is compact in size ...Type I CRISPR-Cas systems utilize the RNA-guided Cascade complex to identify matching DNA targets and the nuclease-helicase Cas3 to degrade them. Among the seven subtypes, type I-C is compact in size and highly active in creating large-sized genome deletions in human cells. Here, we use four cryoelectron microscopy snapshots to define its RNA-guided DNA binding and cleavage mechanisms in high resolution. The non-target DNA strand (NTS) is accommodated by I-C Cascade in a continuous binding groove along the juxtaposed Cas11 subunits. Binding of Cas3 further traps a flexible bulge in NTS, enabling NTS nicking. We identified two anti-CRISPR proteins AcrIC8 and AcrIC9 that strongly inhibit Neisseria lactamica I-C function. Structural analysis showed that AcrIC8 inhibits PAM recognition through allosteric inhibition, whereas AcrIC9 achieves so through direct competition. Both Acrs potently inhibit I-C-mediated genome editing and transcriptional modulation in human cells, providing the first off-switches for type I CRISPR eukaryotic genome engineering. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29877.map.gz emd_29877.map.gz | 40.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29877-v30.xml emd-29877-v30.xml emd-29877.xml emd-29877.xml | 27 KB 27 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_29877.png emd_29877.png | 124.3 KB | ||
| Filedesc metadata |  emd-29877.cif.gz emd-29877.cif.gz | 7.1 KB | ||
| Others |  emd_29877_additional_1.map.gz emd_29877_additional_1.map.gz emd_29877_half_map_1.map.gz emd_29877_half_map_1.map.gz emd_29877_half_map_2.map.gz emd_29877_half_map_2.map.gz | 27.5 MB 40.6 MB 40.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29877 http://ftp.pdbj.org/pub/emdb/structures/EMD-29877 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29877 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29877 | HTTPS FTP |
-Related structure data
| Related structure data |  8g9sMC  8g9tC  8g9uC  8gafC  8gamC  8ganC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_29877.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29877.map.gz / Format: CCP4 / Size: 59.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.4124 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_29877_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_29877_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_29877_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
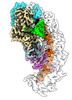
-Entire : Binary complex of AcrIC8 with crRNA bound type I-C Cascade
| Entire | Name: Binary complex of AcrIC8 with crRNA bound type I-C Cascade |
|---|---|
| Components |
|
-Supramolecule #1: Binary complex of AcrIC8 with crRNA bound type I-C Cascade
| Supramolecule | Name: Binary complex of AcrIC8 with crRNA bound type I-C Cascade type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#6 |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 400 KDa |
-Macromolecule #1: Cas7
| Macromolecule | Name: Cas7 / type: protein_or_peptide / ID: 1 / Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 32.208111 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: TIEKRYDFVF LFDVQDGNPN GDPDAGNLPR IDPQTGEGLV TDVCLKRKVR NFIQMTQNDE HHDIFIREKG ILNNLIDEAH EQENVKGKE KGEKTEAARQ YMCSRYYDIR TFGAVMTTGK NAGQVRGPVQ LTFSRSIDPI MTLEHSITRM AVTNEKDASE T GDNRTMGR ...String: TIEKRYDFVF LFDVQDGNPN GDPDAGNLPR IDPQTGEGLV TDVCLKRKVR NFIQMTQNDE HHDIFIREKG ILNNLIDEAH EQENVKGKE KGEKTEAARQ YMCSRYYDIR TFGAVMTTGK NAGQVRGPVQ LTFSRSIDPI MTLEHSITRM AVTNEKDASE T GDNRTMGR KFTVPYGLYR CHGFISTHFA KQTGFSENDL ELFWQALVNM FDHDHSAARG QMNARGLYVF EHSNNLGDAP AD SLFKRIQ VVKKDGVEVV RSFDDYLVSV DDKNLEETKL LRKLGG UniProtKB: UNIPROTKB: A0A378VEU0 |
-Macromolecule #2: Cas11
| Macromolecule | Name: Cas11 / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 14.245184 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GLDRNRQDIG YVLGRLFAVL EKIQAEANPG LNATIADRYF GSASSTPIAV FGTLMRLLPH HLNKLEFEGR AVQLQWEIRQ ILEHCQRFP NHLNLEQQGL FAIGYYHETQ FLFTKDALKN LFNEA UniProtKB: UNIPROTKB: A0A378VF47 |
-Macromolecule #3: Cas5
| Macromolecule | Name: Cas5 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 23.854451 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: RFILEISGDL ACFTRSELKV ERVSYPVITP AAARNILMAI LWKPAIRWKV LKIEILKPIQ WTNIRRNEVG TKMSERSGSL YIEDNRQQR ASMLLKDVAY RIHADFDMTS EAGESDNYVK FAEMFKRRAK KGQYFHQPYL GCREFPCDFR LLEKAEDGLP L EDITQDFG ...String: RFILEISGDL ACFTRSELKV ERVSYPVITP AAARNILMAI LWKPAIRWKV LKIEILKPIQ WTNIRRNEVG TKMSERSGSL YIEDNRQQR ASMLLKDVAY RIHADFDMTS EAGESDNYVK FAEMFKRRAK KGQYFHQPYL GCREFPCDFR LLEKAEDGLP L EDITQDFG FMLYDMDFSK SDPRDSNNAE PMFYQCKAVN GVITVPP UniProtKB: pre-crRNA processing endonuclease |
-Macromolecule #5: Cas8
| Macromolecule | Name: Cas8 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 45.864527 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MILHALTQYY QRKAESAQKG ICLVTGKAAP IARLHNAVKG VNAKPAPFAS VNLSAFESYG KEQGFAFPIG EQAMFEYTTA LNTLLAGEN RFRIGDVTTV CWGAKRTPLE ESLASMINGG GKDKPDEHID AVKTLYKSLY NGQYQKPDGK EKFYLLGLSP N SARIVVRF ...String: MILHALTQYY QRKAESAQKG ICLVTGKAAP IARLHNAVKG VNAKPAPFAS VNLSAFESYG KEQGFAFPIG EQAMFEYTTA LNTLLAGEN RFRIGDVTTV CWGAKRTPLE ESLASMINGG GKDKPDEHID AVKTLYKSLY NGQYQKPDGK EKFYLLGLSP N SARIVVRF WHETTVAALS ESIAAWYDDL QMVRGENSPY PEYMPLPRLL GNLVLDGKME NLPSDLIAQI TDAALNNRVL PV SLLQAAL RRNKAEQKIT YGRASLLKAY INRAIRAGRL KNMKELTMGL DRNRQDIGYV LGRLFAVLEK IQAEANPGLN ATI ADRYFG SASSTPIAVF GTLMRLLPHH LNKLEFEGRA VQLQWEIRQI LEHCQRFPNH LNLEQQGLFA IGYYHETQFL FTKD ALKNL FNEA UniProtKB: UNIPROTKB: A0A378VF47 |
-Macromolecule #6: AcrIC8
| Macromolecule | Name: AcrIC8 / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Rhodobacter phage RcNL1 (virus) Rhodobacter phage RcNL1 (virus) |
| Molecular weight | Theoretical: 8.190161 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SMYAIRKIQF FYGPTDKKSY VGEEAGGRRE LFKTRAEAQA RIEDLEEGVY YLAHNESGRP DYKIVWVRGE |
-Macromolecule #4: RNA (42-MER)
| Macromolecule | Name: RNA (42-MER) / type: rna / ID: 4 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Neisseria lactamica (bacteria) Neisseria lactamica (bacteria) |
| Molecular weight | Theoretical: 13.495021 KDa |
| Sequence | String: AUUGAAACAG GGUCAGCUUG CCGUAGGUGG CAUCGCCCUC GU |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Component - Concentration: 150.0 mM / Component - Formula: NaCl / Component - Name: sodium chloride / Details: 25mM Tris pH 7.5, 150mM NaCl |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Pressure: 0.00039000000000000005 kPa |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 278 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Temperature | Min: 70.0 K / Max: 100.0 K |
| Specialist optics | Energy filter - Name: GIF Bioquantum |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1200 / Number real images: 1200 / Average exposure time: 2.5 sec. / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 3.0 µm / Calibrated defocus min: 1.5 µm / Illumination mode: FLOOD BEAM / Imaging mode: DIFFRACTION / Cs: 2.7 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 2.5 µm / Nominal magnification: 67000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)