+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM Structure of DNPEP | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | DNPEP / aminopeptidase / tetrahedral Symmetry / PEPTIDE BINDING PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報aspartyl aminopeptidase / aminopeptidase activity / metallopeptidase activity / proteolysis / zinc ion binding / cytoplasm 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
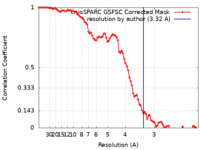
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.32 Å | |||||||||
 データ登録者 データ登録者 | Morgan CE / Yu EW / Zhang Z | |||||||||
| 資金援助 |  米国, 1件 米国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Cell Rep / 年: 2022 ジャーナル: Cell Rep / 年: 2022タイトル: Toward structural-omics of the bovine retinal pigment epithelium. 著者: Christopher E Morgan / Zhemin Zhang / Masaru Miyagi / Marcin Golczak / Edward W Yu /  要旨: The use of an integrated systems biology approach to investigate tissues and organs has been thought to be impracticable in the field of structural biology, where the techniques mainly focus on ...The use of an integrated systems biology approach to investigate tissues and organs has been thought to be impracticable in the field of structural biology, where the techniques mainly focus on determining the structure of a particular biomacromolecule of interest. Here, we report the use of cryoelectron microscopy (cryo-EM) to define the composition of a raw bovine retinal pigment epithelium (RPE) lysate. From this sample, we simultaneously identify and solve cryo-EM structures of seven different RPE enzymes whose functions affect neurotransmitter recycling, iron metabolism, gluconeogenesis, glycolysis, axonal development, and energy homeostasis. Interestingly, dysfunction of these important proteins has been directly linked to several neurodegenerative disorders, including Huntington's disease, amyotrophic lateral sclerosis (ALS), Parkinson's disease, Alzheimer's disease, and schizophrenia. Our work underscores the importance of cryo-EM in facilitating tissue and organ proteomics at the atomic level. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_26350.map.gz emd_26350.map.gz | 14.8 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-26350-v30.xml emd-26350-v30.xml emd-26350.xml emd-26350.xml | 19.8 KB 19.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_26350_fsc.xml emd_26350_fsc.xml | 8.4 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_26350.png emd_26350.png | 145.5 KB | ||
| Filedesc metadata |  emd-26350.cif.gz emd-26350.cif.gz | 5.9 KB | ||
| その他 |  emd_26350_additional_1.map.gz emd_26350_additional_1.map.gz emd_26350_half_map_1.map.gz emd_26350_half_map_1.map.gz emd_26350_half_map_2.map.gz emd_26350_half_map_2.map.gz | 32.3 MB 59.2 MB 59.2 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26350 http://ftp.pdbj.org/pub/emdb/structures/EMD-26350 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26350 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26350 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7u5hMC  7u5iC  7u5jC  7u5kC  7u5lC  7u5mC  7u5nC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_26350.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_26350.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
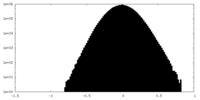
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-追加マップ: #1
| ファイル | emd_26350_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
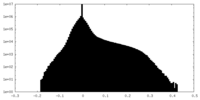
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_26350_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
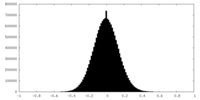
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_26350_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
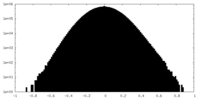
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Dodecamer of Aspartyl Aminopeptidase
| 全体 | 名称: Dodecamer of Aspartyl Aminopeptidase |
|---|---|
| 要素 |
|
-超分子 #1: Dodecamer of Aspartyl Aminopeptidase
| 超分子 | 名称: Dodecamer of Aspartyl Aminopeptidase / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1 |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: Aspartyl aminopeptidase
| 分子 | 名称: Aspartyl aminopeptidase / タイプ: protein_or_peptide / ID: 1 / コピー数: 12 / 光学異性体: LEVO / EC番号: aspartyl aminopeptidase |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 51.893188 KDa |
| 配列 | 文字列: MSGRARKEAV QAAARELLKF VNRSPSPFHA VAECRSRLLQ AGFHELKETE SWDIKPESKY FLTRNSSTII AFAVGGQYVP GNGFSLIGA HTDSPCLRVK RRSRRSQVGF QQVGVETYGG GIWSTWFDRD LTLAGRVIVK CPTSGRLEQR LVHVDRPILR I PHLAIHLQ ...文字列: MSGRARKEAV QAAARELLKF VNRSPSPFHA VAECRSRLLQ AGFHELKETE SWDIKPESKY FLTRNSSTII AFAVGGQYVP GNGFSLIGA HTDSPCLRVK RRSRRSQVGF QQVGVETYGG GIWSTWFDRD LTLAGRVIVK CPTSGRLEQR LVHVDRPILR I PHLAIHLQ RNVNENFGPN MEMHLVPILA TSIQEELEKG TPEPGPLNAT DERHHSVLTS LLCAHLGLSP EDILEMELCL AD TQPAVLG GAYEEFIFAP RLDNLHSCFC ALQALIDSCS APASLAADPH VRMIALYDNE EVGSESAQGA QSLLTELVLR RIS ASPQHL TAFEEAIPKS YMISADMAHA VHPNYLDKHE ENHRPLFHKG PVIKVNSKQR YASNAVSEAL IREVASSVGV PLQD LMVRN DSPCGTTIGP ILASRLGLRV LDLGSPQLAM HSIRETACTT GVLQTITLFK GFFELFPSLS RSLLVD UniProtKB: Aspartyl aminopeptidase |
-分子 #2: ZINC ION
| 分子 | 名称: ZINC ION / タイプ: ligand / ID: 2 / コピー数: 24 / 式: ZN |
|---|---|
| 分子量 | 理論値: 65.409 Da |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 平均電子線量: 37.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: SPOT SCAN / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 1.0 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)