+ Open data
Open data
- Basic information
Basic information
| Entry | 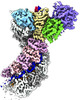 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cascade complex from type I-A CRISPR-Cas system | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
| Function / homology |  Function and homology information Function and homology informationexonuclease activity / endonuclease activity / defense response to virus / nucleic acid binding / RNA helicase activity / Hydrolases; Acting on ester bonds / hydrolase activity / ATP binding / metal ion binding / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Geobacter sulfurreducens (bacteria) / Geobacter sulfurreducens (bacteria) /   Pyrococcus furiosus DSM 3638 (archaea) / Pyrococcus furiosus DSM 3638 (archaea) /  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||
 Authors Authors | Hu C / Ni D / Nam KH / Majumdar S / McLean J / Stahlberg H / Terns M / Ke A | ||||||||||||
| Funding support |  United States, United States,  Switzerland, 3 items Switzerland, 3 items
| ||||||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2022 Journal: Mol Cell / Year: 2022Title: Allosteric control of type I-A CRISPR-Cas3 complexes and establishment as effective nucleic acid detection and human genome editing tools. Authors: Chunyi Hu / Dongchun Ni / Ki Hyun Nam / Sonali Majumdar / Justin McLean / Henning Stahlberg / Michael P Terns / Ailong Ke /    Abstract: Type I CRISPR-Cas systems typically rely on a two-step process to degrade DNA. First, an RNA-guided complex named Cascade identifies the complementary DNA target. The helicase-nuclease fusion enzyme ...Type I CRISPR-Cas systems typically rely on a two-step process to degrade DNA. First, an RNA-guided complex named Cascade identifies the complementary DNA target. The helicase-nuclease fusion enzyme Cas3 is then recruited in trans for processive DNA degradation. Contrary to this model, here, we show that type I-A Cascade and Cas3 function as an integral effector complex. We provide four cryoelectron microscopy (cryo-EM) snapshots of the Pyrococcus furiosus (Pfu) type I-A effector complex in different stages of DNA recognition and degradation. The HD nuclease of Cas3 is autoinhibited inside the effector complex. It is only allosterically activated upon full R-loop formation, when the entire targeted region has been validated by the RNA guide. The mechanistic insights inspired us to convert Pfu Cascade-Cas3 into a high-sensitivity, low-background, and temperature-activated nucleic acid detection tool. Moreover, Pfu CRISPR-Cas3 shows robust bi-directional deletion-editing activity in human cells, which could find usage in allele-specific inactivation of disease-causing mutations. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26084.map.gz emd_26084.map.gz | 52 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26084-v30.xml emd-26084-v30.xml emd-26084.xml emd-26084.xml | 26.6 KB 26.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_26084.png emd_26084.png | 154.6 KB | ||
| Others |  emd_26084_additional_1.map.gz emd_26084_additional_1.map.gz emd_26084_additional_2.map.gz emd_26084_additional_2.map.gz | 97.3 MB 97.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26084 http://ftp.pdbj.org/pub/emdb/structures/EMD-26084 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26084 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26084 | HTTPS FTP |
-Validation report
| Summary document |  emd_26084_validation.pdf.gz emd_26084_validation.pdf.gz | 188.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_26084_full_validation.pdf.gz emd_26084_full_validation.pdf.gz | 188.2 KB | Display | |
| Data in XML |  emd_26084_validation.xml.gz emd_26084_validation.xml.gz | 503 B | Display | |
| Data in CIF |  emd_26084_validation.cif.gz emd_26084_validation.cif.gz | 374 B | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26084 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26084 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26084 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26084 | HTTPS FTP |
-Related structure data
| Related structure data |  7traMC  7tr6C  7tr8C  7tr9C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26084.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26084.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.24 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Cas3 part local refinement
| File | emd_26084_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cas3 part local refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Sharpened map
| File | emd_26084_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : full R-loop state of Cas3-Cascade complex from Pyrococcus furiosu...
+Supramolecule #1: full R-loop state of Cas3-Cascade complex from Pyrococcus furiosu...
+Macromolecule #1: CRISPR-associated helicase Cas3
+Macromolecule #2: Cas8a
+Macromolecule #3: Cas11a
+Macromolecule #4: Cas7a
+Macromolecule #5: Cas5a
+Macromolecule #6: CRISPR-associated endonuclease Cas3-HD
+Macromolecule #7: crRNA (44-MER)
+Macromolecule #8: Target strand DNA (44-MER)
+Macromolecule #9: Non-Target strand DNA (25-MER)
+Macromolecule #10: NICKEL (II) ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Component - Concentration: 150.0 mM / Component - Formula: NaCl / Component - Name: sodium chloride |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV / Details: 6 seconds. |
- Electron microscopy
Electron microscopy
| Microscope | TFS TALOS |
|---|---|
| Specialist optics | Phase plate: VOLTA PHASE PLATE |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number real images: 1209 / Average exposure time: 2.5 sec. / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus min: 1.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: DIFFRACTION / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: OTHER |
|---|---|
| Output model |  PDB-7tra: |
 Movie
Movie Controller
Controller





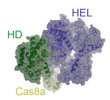

 Z
Z Y
Y X
X


















