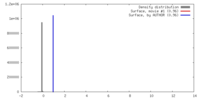
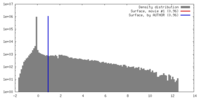
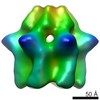
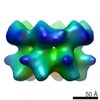
登録情報 データベース : EMDB / ID : EMD-2590タイトル Inter-ring rotations of AAA ATPase p97 Reconstruction of p97 in presence of ADP, conformation 1 試料 : p97 in presence of ADP, conformation 1タンパク質・ペプチド : Transitional endoplasmic reticulum ATPase / 機能・相同性 分子機能 ドメイン・相同性 構成要素
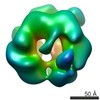
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Mus musculus (ハツカネズミ)手法 / / 解像度 : 20.0 Å Yeung HO / Forster A / Bebeacua C / Niwa H / Ewens C / McKeown C / Zhang X / Freemont PS ジャーナル : Open Biol / 年 : 2014タイトル : Inter-ring rotations of AAA ATPase p97 revealed by electron cryomicroscopy.著者 : Heidi O Yeung / Andreas Förster / Cecilia Bebeacua / Hajime Niwa / Caroline Ewens / Ciarán McKeown / Xiaodong Zhang / Paul S Freemont / 要旨 : The type II AAA+ protein p97 is involved in numerous cellular activities, including endoplasmic reticulum-associated degradation, transcription activation, membrane fusion and cell-cycle control. ... The type II AAA+ protein p97 is involved in numerous cellular activities, including endoplasmic reticulum-associated degradation, transcription activation, membrane fusion and cell-cycle control. These activities are at least in part regulated by the ubiquitin system, in which p97 is thought to target ubiquitylated protein substrates within macromolecular complexes and assist in their extraction or disassembly. Although ATPase activity is essential for p97 function, little is known about how ATP binding or hydrolysis is coupled with p97 conformational changes and substrate remodelling. Here, we have used single-particle electron cryomicroscopy (cryo-EM) to study the effect of nucleotides on p97 conformation. We have identified conformational heterogeneity within the cryo-EM datasets from which we have resolved two major p97 conformations. A comparison of conformations reveals inter-ring rotations upon nucleotide binding and hydrolysis that may be linked to the remodelling of target protein complexes. 履歴 登録 2014年2月21日 - ヘッダ(付随情報) 公開 2014年2月26日 - マップ公開 2014年2月26日 - 更新 2014年3月19日 - 現状 2014年3月19日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 引用
引用 ジャーナル: Open Biol / 年: 2014
ジャーナル: Open Biol / 年: 2014
 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_2590.map.gz
emd_2590.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-2590-v30.xml
emd-2590-v30.xml emd-2590.xml
emd-2590.xml EMDBヘッダ
EMDBヘッダ emd_2590.jpg
emd_2590.jpg http://ftp.pdbj.org/pub/emdb/structures/EMD-2590
http://ftp.pdbj.org/pub/emdb/structures/EMD-2590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2590
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2590 emd_2590_validation.pdf.gz
emd_2590_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_2590_full_validation.pdf.gz
emd_2590_full_validation.pdf.gz emd_2590_validation.xml.gz
emd_2590_validation.xml.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2590
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2590 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2590
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2590 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_2590.map.gz / 形式: CCP4 / 大きさ: 3.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_2590.map.gz / 形式: CCP4 / 大きさ: 3.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素

 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN 画像解析
画像解析 ムービー
ムービー コントローラー
コントローラー




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)