[English] 日本語
 Yorodumi
Yorodumi- EMDB-21895: Monomer yeast ATP synthase (F1Fo) reconstituted in nanodisc with ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-21895 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Monomer yeast ATP synthase (F1Fo) reconstituted in nanodisc with inhibitor of bedaquiline bound | |||||||||
 Map data Map data | Monomer yeast ATP synthase (F1Fo) reconstituted in nanodisc with inhibitor of bedaquiline bound | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Luo M / Liao MF / Mueller DM | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2020 Journal: Commun Biol / Year: 2020Title: Bedaquiline inhibits the yeast and human mitochondrial ATP synthases. Authors: Min Luo / Wenchang Zhou / Hiral Patel / Anurag P Srivastava / Jindrich Symersky / Michał M Bonar / José D Faraldo-Gómez / Maofu Liao / David M Mueller /  Abstract: Bedaquiline (BDQ, Sirturo) has been approved to treat multidrug resistant forms of Mycobacterium tuberculosis. Prior studies suggested that BDQ was a selective inhibitor of the ATP synthase from M. ...Bedaquiline (BDQ, Sirturo) has been approved to treat multidrug resistant forms of Mycobacterium tuberculosis. Prior studies suggested that BDQ was a selective inhibitor of the ATP synthase from M. tuberculosis. However, Sirturo treatment leads to an increased risk of cardiac arrhythmias and death, raising the concern that this adverse effect results from inhibition at a secondary site. Here we show that BDQ is a potent inhibitor of the yeast and human mitochondrial ATP synthases. Single-particle cryo-EM reveals that the site of BDQ inhibition partially overlaps with that of the inhibitor oligomycin. Molecular dynamics simulations indicate that the binding mode of BDQ to this site is similar to that previously seen for a mycobacterial enzyme, explaining the observed lack of selectivity. We propose that derivatives of BDQ ought to be made to increase its specificity toward the mycobacterial enzyme and thereby reduce the side effects for patients that are treated with Sirturo. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_21895.map.gz emd_21895.map.gz | 112.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-21895-v30.xml emd-21895-v30.xml emd-21895.xml emd-21895.xml | 12.7 KB 12.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_21895.png emd_21895.png | 64.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21895 http://ftp.pdbj.org/pub/emdb/structures/EMD-21895 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21895 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21895 | HTTPS FTP |
-Validation report
| Summary document |  emd_21895_validation.pdf.gz emd_21895_validation.pdf.gz | 78.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_21895_full_validation.pdf.gz emd_21895_full_validation.pdf.gz | 77.5 KB | Display | |
| Data in XML |  emd_21895_validation.xml.gz emd_21895_validation.xml.gz | 494 B | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21895 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21895 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21895 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21895 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_21895.map.gz / Format: CCP4 / Size: 122.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_21895.map.gz / Format: CCP4 / Size: 122.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Monomer yeast ATP synthase (F1Fo) reconstituted in nanodisc with inhibitor of bedaquiline bound | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.235 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
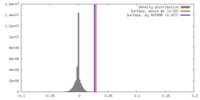
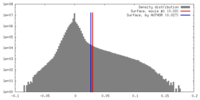
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Monomer yeast ATP synthase Fo reconstituted in nanodisc generated...
| Entire | Name: Monomer yeast ATP synthase Fo reconstituted in nanodisc generated in the presence of Bedaquiline |
|---|---|
| Components |
|
-Supramolecule #1: Monomer yeast ATP synthase Fo reconstituted in nanodisc generated...
| Supramolecule | Name: Monomer yeast ATP synthase Fo reconstituted in nanodisc generated in the presence of Bedaquiline type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#8 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL |
|---|---|
| Buffer | pH: 8 / Details: 20 mM Tris-HCl, 150 mM NaCl, pH 8.0 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 91 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Temperature | Min: 80.0 K / Max: 105.0 K |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number real images: 9258 / Average electron dose: 8.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.8 µm / Calibrated defocus min: 0.8 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal magnification: 31000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: |
|---|---|
| Refinement | Space: REAL |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)