[English] 日本語
 Yorodumi
Yorodumi- EMDB-18999: Cryo-EM structure of coagulation factor beta-XIIa in complex with... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of coagulation factor beta-XIIa in complex with the garadacimab Fab fragment (symmetric dimer) | |||||||||
 Map data Map data | Sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Complex / coagulation / trypsin-like serine protease / hereditary angioedema (HAE) / BLOOD CLOTTING | |||||||||
| Function / homology |  Function and homology information Function and homology informationcoagulation factor XIIa / plasma kallikrein-kinin cascade / Factor XII activation / Defective SERPING1 causes hereditary angioedema / response to misfolded protein / positive regulation of plasminogen activation / blood coagulation, intrinsic pathway / misfolded protein binding / positive regulation of fibrinolysis / zymogen activation ...coagulation factor XIIa / plasma kallikrein-kinin cascade / Factor XII activation / Defective SERPING1 causes hereditary angioedema / response to misfolded protein / positive regulation of plasminogen activation / blood coagulation, intrinsic pathway / misfolded protein binding / positive regulation of fibrinolysis / zymogen activation / Defective factor XII causes hereditary angioedema / protein autoprocessing / positive regulation of blood coagulation / rough endoplasmic reticulum / fibrinolysis / Intrinsic Pathway of Fibrin Clot Formation / protein processing / blood coagulation / collagen-containing extracellular matrix / serine-type endopeptidase activity / innate immune response / calcium ion binding / extracellular space / extracellular exosome / extracellular region / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
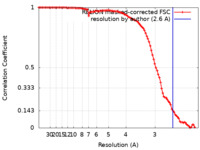
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Drulyte I | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Structural basis for inhibition of beta-FXIIa by garadacimab Authors: Drulyte I / Ghai R / Ow SY / Kapp EA / Quek AJ / Panousis C / Wilson MJ / Nash AD / Pelzing M | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_18999.map.gz emd_18999.map.gz | 79 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-18999-v30.xml emd-18999-v30.xml emd-18999.xml emd-18999.xml | 23.6 KB 23.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_18999_fsc.xml emd_18999_fsc.xml | 9.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_18999.png emd_18999.png | 91.2 KB | ||
| Masks |  emd_18999_msk_1.map emd_18999_msk_1.map emd_18999_msk_2.map emd_18999_msk_2.map | 83.7 MB 83.7 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-18999.cif.gz emd-18999.cif.gz | 7 KB | ||
| Others |  emd_18999_additional_1.map.gz emd_18999_additional_1.map.gz emd_18999_additional_2.map.gz emd_18999_additional_2.map.gz emd_18999_half_map_1.map.gz emd_18999_half_map_1.map.gz emd_18999_half_map_2.map.gz emd_18999_half_map_2.map.gz | 42.4 MB 1.9 MB 77.5 MB 77.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-18999 http://ftp.pdbj.org/pub/emdb/structures/EMD-18999 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18999 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-18999 | HTTPS FTP |
-Validation report
| Summary document |  emd_18999_validation.pdf.gz emd_18999_validation.pdf.gz | 847.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_18999_full_validation.pdf.gz emd_18999_full_validation.pdf.gz | 847.1 KB | Display | |
| Data in XML |  emd_18999_validation.xml.gz emd_18999_validation.xml.gz | 17.1 KB | Display | |
| Data in CIF |  emd_18999_validation.cif.gz emd_18999_validation.cif.gz | 22.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18999 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18999 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18999 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-18999 | HTTPS FTP |
-Related structure data
| Related structure data |  8r8dMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_18999.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_18999.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.095 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_18999_msk_1.map emd_18999_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Mask #2
| File |  emd_18999_msk_2.map emd_18999_msk_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened map
| File | emd_18999_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Local resolution-filtered map
| File | emd_18999_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Local resolution-filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map A
| File | emd_18999_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map B
| File | emd_18999_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Ternary complex of coagulation factor beta-XIIa with garadacimab ...
| Entire | Name: Ternary complex of coagulation factor beta-XIIa with garadacimab Fab fragment and anti-LC-lambda VHH |
|---|---|
| Components |
|
-Supramolecule #1: Ternary complex of coagulation factor beta-XIIa with garadacimab ...
| Supramolecule | Name: Ternary complex of coagulation factor beta-XIIa with garadacimab Fab fragment and anti-LC-lambda VHH type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 174 KDa |
-Macromolecule #1: Coagulation factor XII
| Macromolecule | Name: Coagulation factor XII / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 26.53775 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: VVGGLVALRG AHPYIAALYW GHSFCAGSLI APCWVLTAAH CLQDRPAPED LTVVLGQERR NHSCEPCQTL AVRSYRLHEA FSPVSYQHD LALLRLQEDA DGSCALLSPY VQPVSLPSGA ARPSETTLCQ VAGWGHQFEG AEEYASFLQE AQVPFLSLER C SAPDVHGS ...String: VVGGLVALRG AHPYIAALYW GHSFCAGSLI APCWVLTAAH CLQDRPAPED LTVVLGQERR NHSCEPCQTL AVRSYRLHEA FSPVSYQHD LALLRLQEDA DGSCALLSPY VQPVSLPSGA ARPSETTLCQ VAGWGHQFEG AEEYASFLQE AQVPFLSLER C SAPDVHGS SILPGMLCAG FLEGGTDACQ GDSGGPLVCE DQAAERRLTL QGIISWGSGC GDRNKPGVYT DVAYYLAWIR EH TVSEPEA UniProtKB: Coagulation factor XII |
-Macromolecule #2: Garadacimab heavy chain variable region
| Macromolecule | Name: Garadacimab heavy chain variable region / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 25.120281 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: EVQLLESGGG LVQPGGSLRL SCAASGFTFS KYIMQWVRQA PGKGLEWVSG IDIPTKGTVY ADSVKGRFTI SRDNSKNTLY LQMNSLRAE DTAVYYCARA LPRSGYLISP HYYYYALDVW GQGTTVTVSS ASTKGPSVFP LAPCSRSTSE STAALGCLVK D YFPEPVTV ...String: EVQLLESGGG LVQPGGSLRL SCAASGFTFS KYIMQWVRQA PGKGLEWVSG IDIPTKGTVY ADSVKGRFTI SRDNSKNTLY LQMNSLRAE DTAVYYCARA LPRSGYLISP HYYYYALDVW GQGTTVTVSS ASTKGPSVFP LAPCSRSTSE STAALGCLVK D YFPEPVTV SWNSGALTSG VHTFPAVLQS SGLYSLSSVV TVPSSSLGTK TYTCNVDHKP SNTKVDKRVE SKYGPP |
-Macromolecule #3: Garadacimab light chain variable region
| Macromolecule | Name: Garadacimab light chain variable region / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 22.762174 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QSVLTQPPSA SGTPGQRVTI SCSGSSSNIG RNYVYWYQQL PGTAPKLLIY SNNQRPSGVP DRFSGSKSGT SASLAISGLR SEDEADYYC AAWDASLRGV FGGGTKLTVL GQPKAAPSVT LFPPSSEELQ ANKATLVCLI SDFYPGAVTV AWKADSSPVK A GVETTTPS ...String: QSVLTQPPSA SGTPGQRVTI SCSGSSSNIG RNYVYWYQQL PGTAPKLLIY SNNQRPSGVP DRFSGSKSGT SASLAISGLR SEDEADYYC AAWDASLRGV FGGGTKLTVL GQPKAAPSVT LFPPSSEELQ ANKATLVCLI SDFYPGAVTV AWKADSSPVK A GVETTTPS KQSNNKYAAS SYLSLTPEQW KSHRSYSCQV THEGSTVEKT VAPTECS |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 18 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL |
|---|---|
| Buffer | pH: 5.5 / Details: 10 mM Na Acetate, 100 mM NaCl pH 5.5 |
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR / Details: plasma current 20 mA |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
| Details | Immediately before blotting and plunge freezing, fluorinated octyl maltoside (FOM) was added to the sample to the final concentration of 0.005%-0.01% (w/v) |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: TFS Selectris / Energy filter - Slit width: 10 eV Details: Electron source E-CFEG (cold-FEG), energy filter Selectris X |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Number grids imaged: 2 / Number real images: 4000 / Average electron dose: 50.0 e/Å2 / Details: 2000 with 0.005% FOM and 2000 with 0.01% FOM |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.25 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 165000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z
Z Y
Y X
X