[English] 日本語
 Yorodumi
Yorodumi- EMDB-15277: Single Particle cryo-EM of the lipid binding protein P116 (MPN213... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Single Particle cryo-EM of the lipid binding protein P116 (MPN213) from Mycoplasma pneumoniae bound to HDL. | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
| Biological species |  Mycoplasma pneumoniae M129 (bacteria) Mycoplasma pneumoniae M129 (bacteria) | ||||||||||||
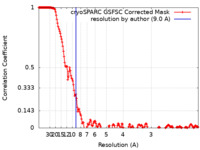
| Method | single particle reconstruction / cryo EM / Resolution: 9.0 Å | ||||||||||||
 Authors Authors | Sprankel L / Vizarraga D | ||||||||||||
| Funding support |  Germany, 3 items Germany, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Essential protein P116 extracts cholesterol and other indispensable lipids for Mycoplasmas. Authors: Lasse Sprankel / David Vizarraga / Jesús Martín / Sina Manger / Jakob Meier-Credo / Marina Marcos / Josep Julve / Noemi Rotllan / Margot P Scheffer / Joan Carles Escolà-Gil / Julian D ...Authors: Lasse Sprankel / David Vizarraga / Jesús Martín / Sina Manger / Jakob Meier-Credo / Marina Marcos / Josep Julve / Noemi Rotllan / Margot P Scheffer / Joan Carles Escolà-Gil / Julian D Langer / Jaume Piñol / Ignacio Fita / Achilleas S Frangakis /   Abstract: Mycoplasma pneumoniae, responsible for approximately 30% of community-acquired human pneumonia, needs to extract lipids from the host environment for survival and proliferation. Here, we report a ...Mycoplasma pneumoniae, responsible for approximately 30% of community-acquired human pneumonia, needs to extract lipids from the host environment for survival and proliferation. Here, we report a comprehensive structural and functional analysis of the previously uncharacterized protein P116 (MPN_213). Single-particle cryo-electron microscopy of P116 reveals a homodimer presenting a previously unseen fold, forming a huge hydrophobic cavity, which is fully accessible to solvent. Lipidomics analysis shows that P116 specifically extracts lipids such as phosphatidylcholine, sphingomyelin and cholesterol. Structures of different conformational states reveal the mechanism by which lipids are extracted. This finding immediately suggests a way to control Mycoplasma infection by interfering with lipid uptake. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15277.map.gz emd_15277.map.gz | 483.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15277-v30.xml emd-15277-v30.xml emd-15277.xml emd-15277.xml | 15.9 KB 15.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_15277_fsc.xml emd_15277_fsc.xml | 17.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_15277.png emd_15277.png | 13.6 KB | ||
| Masks |  emd_15277_msk_1.map emd_15277_msk_1.map | 512 MB |  Mask map Mask map | |
| Others |  emd_15277_half_map_1.map.gz emd_15277_half_map_1.map.gz emd_15277_half_map_2.map.gz emd_15277_half_map_2.map.gz | 475.8 MB 475.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15277 http://ftp.pdbj.org/pub/emdb/structures/EMD-15277 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15277 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15277 | HTTPS FTP |
-Validation report
| Summary document |  emd_15277_validation.pdf.gz emd_15277_validation.pdf.gz | 691.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_15277_full_validation.pdf.gz emd_15277_full_validation.pdf.gz | 690.8 KB | Display | |
| Data in XML |  emd_15277_validation.xml.gz emd_15277_validation.xml.gz | 26.4 KB | Display | |
| Data in CIF |  emd_15277_validation.cif.gz emd_15277_validation.cif.gz | 34.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15277 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15277 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15277 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-15277 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_15277.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15277.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.05 Å | ||||||||||||||||||||||||||||||||||||
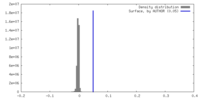
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_15277_msk_1.map emd_15277_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
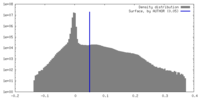
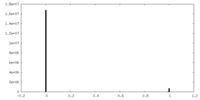
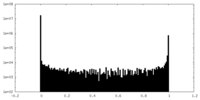
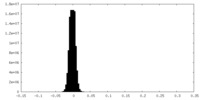
| Density Histograms |
-Half map: #1
| File | emd_15277_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
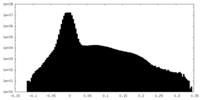
| Density Histograms |
-Half map: #2
| File | emd_15277_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
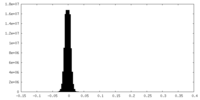
| Density Histograms |
- Sample components
Sample components
-Entire : P116 homodimer mixed with HDL
| Entire | Name: P116 homodimer mixed with HDL |
|---|---|
| Components |
|
-Supramolecule #1: P116 homodimer mixed with HDL
| Supramolecule | Name: P116 homodimer mixed with HDL / type: complex / ID: 1 / Chimera: Yes / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Mycoplasma pneumoniae M129 (bacteria) Mycoplasma pneumoniae M129 (bacteria) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.25 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Component - Concentration: 20.0 mM / Component - Name: Tris |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
| Details | 250 ug/ml empty P116 was mixed with 1160 ug/ml HDL |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum SE / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3710 pixel / Digitization - Dimensions - Height: 3838 pixel / Number real images: 3114 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Calibrated defocus max: 3.5 µm / Calibrated defocus min: 1.0 µm / Calibrated magnification: 130000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)