+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12273 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the human FERRY complex | |||||||||
 Map data Map data | Cryo-EM structure of the FERRY complex core | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Human FERRY complex / Five-subunit Early endosome RNA and Ribosome intermediarY complex / Intracellular RNA transport / Early Endosome-associated transport of RNA / RNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of intracellular mRNA localization / glyoxalase III activity / : / early endosome membrane / protein-macromolecule adaptor activity / early endosome / oxidoreductase activity / mRNA binding / RNA binding / extracellular exosome ...regulation of intracellular mRNA localization / glyoxalase III activity / : / early endosome membrane / protein-macromolecule adaptor activity / early endosome / oxidoreductase activity / mRNA binding / RNA binding / extracellular exosome / membrane / identical protein binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Quentin D / Klink BU / Raunser S | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Structural basis of mRNA binding by the human FERRY Rab5 effector complex. Authors: Dennis Quentin / Jan S Schuhmacher / Björn U Klink / Jeni Lauer / Tanvir R Shaikh / Pim J Huis In 't Veld / Luisa M Welp / Henning Urlaub / Marino Zerial / Stefan Raunser /  Abstract: The pentameric FERRY Rab5 effector complex is a molecular link between mRNA and early endosomes in mRNA intracellular distribution. Here, we determine the cryo-EM structure of human FERRY. It reveals ...The pentameric FERRY Rab5 effector complex is a molecular link between mRNA and early endosomes in mRNA intracellular distribution. Here, we determine the cryo-EM structure of human FERRY. It reveals a unique clamp-like architecture that bears no resemblance to any known structure of Rab effectors. A combination of functional and mutational studies reveals that while the Fy-2 C-terminal coiled-coil acts as binding region for Fy-1/3 and Rab5, both coiled-coils and Fy-5 concur to bind mRNA. Mutations causing truncations of Fy-2 in patients with neurological disorders impair Rab5 binding or FERRY complex assembly. Thus, Fy-2 serves as a binding hub connecting all five complex subunits and mediating the binding to mRNA and early endosomes via Rab5. Our study provides mechanistic insights into long-distance mRNA transport and demonstrates that the particular architecture of FERRY is closely linked to a previously undescribed mode of RNA binding, involving coiled-coil domains. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12273.map.gz emd_12273.map.gz | 4.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12273-v30.xml emd-12273-v30.xml emd-12273.xml emd-12273.xml | 21.5 KB 21.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_12273.png emd_12273.png | 106.3 KB | ||
| Filedesc metadata |  emd-12273.cif.gz emd-12273.cif.gz | 7.5 KB | ||
| Others |  emd_12273_additional_1.map.gz emd_12273_additional_1.map.gz | 66.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12273 http://ftp.pdbj.org/pub/emdb/structures/EMD-12273 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12273 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12273 | HTTPS FTP |
-Related structure data
| Related structure data |  7nd2MC  8a3oC  8a3pC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12273.map.gz / Format: CCP4 / Size: 70.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12273.map.gz / Format: CCP4 / Size: 70.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of the FERRY complex core | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
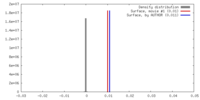
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: L-AFTER filtered Cryo-EM density map of the FERRY complex core
| File | emd_12273_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | L-AFTER filtered Cryo-EM density map of the FERRY complex core | ||||||||||||
| Projections & Slices |
| ||||||||||||
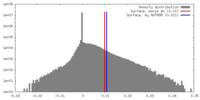
| Density Histograms |
- Sample components
Sample components
-Entire : Human FERRY (Five-subunit Early endosome RNA and Ribosome interme...
| Entire | Name: Human FERRY (Five-subunit Early endosome RNA and Ribosome intermediarY) complex |
|---|---|
| Components |
|
-Supramolecule #1: Human FERRY (Five-subunit Early endosome RNA and Ribosome interme...
| Supramolecule | Name: Human FERRY (Five-subunit Early endosome RNA and Ribosome intermediarY) complex type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Supramolecule #2: Protein phosphatase 1 regulatory subunit 21
| Supramolecule | Name: Protein phosphatase 1 regulatory subunit 21 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: Quinone oxidoreductase-like protein 1
| Supramolecule | Name: Quinone oxidoreductase-like protein 1 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #4: Glutamine amidotransferase-like class 1 domain-containing protein 1
| Supramolecule | Name: Glutamine amidotransferase-like class 1 domain-containing protein 1 type: complex / ID: 4 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Protein phosphatase 1 regulatory subunit 21
| Macromolecule | Name: Protein phosphatase 1 regulatory subunit 21 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 88.782836 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAAAMASAEL QGKYQKLAQE YSKLRAQNQV LKKGVVDEQA NSAALKEQLK MKDQSLRKLQ QEMDSLTFRN LQLAKRVELL QDELALSEP RGKKNKKSGE SSSQLSQEQK SVFDEDLQKK IEENERLHIQ FFEADEQHKH VEAELRSRLA TLETEAAQHQ A VVDGLTRK ...String: MAAAMASAEL QGKYQKLAQE YSKLRAQNQV LKKGVVDEQA NSAALKEQLK MKDQSLRKLQ QEMDSLTFRN LQLAKRVELL QDELALSEP RGKKNKKSGE SSSQLSQEQK SVFDEDLQKK IEENERLHIQ FFEADEQHKH VEAELRSRLA TLETEAAQHQ A VVDGLTRK YMETIEKLQN DKAKLEVKSQ TLEKEAKECR LRTEECQLQL KTLHEDLSGR LEESLSIINE KVPFNDTKYS QY NALNVPL HNRRHQLKMR DIAGQALAFV QDLVTALLNF HTYTEQRIQI FPVDSAIDTI SPLNQKFSQY LHENASYVRP LEE GMLHLF ESITEDTVTV LETTVKLKTF SEHLTSYICF LRKILPYQLK SLEEECESSL CTSALRARNL ELSQDMKKMT AVFE KLQTY IALLALPSTE PDGLLRTNYS SVLTNVGAAL HGFHDVMKDI SKHYSQKAAI EHELPTATQK LITTNDCILS SVVAL TNGA GKIASFFSNN LDYFIASLSY GPKAASGFIS PLSAECMLQY KKKAAAYMKS LRKPLLESVP YEEALANRRI LLSSTE SRE GLAQQVQQSL EKISKLEQEK EHWMLEAQLA KIKLEKENQR IADKLKNTGS AQLVGLAQEN AAVSNTAGQD EATAKAV LE PIQSTSLIGT LTRTSDSEVP DVESREDLIK NHYMARIVEL TSQLQLADSK SVHFYAECRA LSKRLALAEK SKEALTEE M KLASQNISRL QDELTTTKRS YEDQLSMMSD HLCSMNETLS KQREEIDTLK MSSKGNSKKN KSR UniProtKB: Protein phosphatase 1 regulatory subunit 21 |
-Macromolecule #2: Quinone oxidoreductase-like protein 1
| Macromolecule | Name: Quinone oxidoreductase-like protein 1 / type: protein_or_peptide / ID: 2 / Details: N-terminal His-6 tag / Number of copies: 2 / Enantiomer: LEVO / EC number: Oxidoreductases |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.661414 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSHHHHHHKG LYFQQSSTDE EITFVFQEKE DLPVTEDNFV KLQVKACALS QINTKLLAEM KMKKDLFPVG REIAGIVLDV GSKVSFFQP DDEVVGILPL DSEDPGLCEV VRVHEHYLVH KPEKVTWTEA AGSIRDGVRA YTALHYLSHL SPGKSVLIMD G ASAFGTIA ...String: MSHHHHHHKG LYFQQSSTDE EITFVFQEKE DLPVTEDNFV KLQVKACALS QINTKLLAEM KMKKDLFPVG REIAGIVLDV GSKVSFFQP DDEVVGILPL DSEDPGLCEV VRVHEHYLVH KPEKVTWTEA AGSIRDGVRA YTALHYLSHL SPGKSVLIMD G ASAFGTIA IQLAHHRGAK VISTACSLED KQCLERFRPP IARVIDVSNG KVHVAESCLE ETGGLGVDIV LDAGVRLYSK DD EPAVKLQ LLPHKHDIIT LLGVGGHWVT TEENLQLDPP DSHCLFLKGA TLAFLNDEVW NLSNVQQGKY LCILKDVMEK LST GVFRPQ LDEPIPLYEA KVSMEAVQKN QGRKKQVVQF UniProtKB: Ferry endosomal RAB5 effector complex subunit 4 |
-Macromolecule #3: Glutamine amidotransferase-like class 1 domain-containing protein 1
| Macromolecule | Name: Glutamine amidotransferase-like class 1 domain-containing protein 1 type: protein_or_peptide / ID: 3 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 24.237488 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSHHHHHHAS ERLPNRPACL LVASGAAEGV SAQSFLHCFT MASTAFNLQV ATPGGKAMEF VDVTESNARW VQDFRLKAYA SPAKLESID GARYHALLIP SCPGALTDLA SSGSLARILQ HFHSESKPIC AVGHGVAALC CATNEDRSWV FDSYSLTGPS V CELVRAPG ...String: MSHHHHHHAS ERLPNRPACL LVASGAAEGV SAQSFLHCFT MASTAFNLQV ATPGGKAMEF VDVTESNARW VQDFRLKAYA SPAKLESID GARYHALLIP SCPGALTDLA SSGSLARILQ HFHSESKPIC AVGHGVAALC CATNEDRSWV FDSYSLTGPS V CELVRAPG FARLPLVVED FVKDSGACFS ASEPDAVHVV LDRHLVTGQN ASSTVPAVQN LLFLCGSRK UniProtKB: Glutamine amidotransferase-like class 1 domain-containing protein 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.7 mg/mL | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 40 sec. / Pretreatment - Atmosphere: AIR | ||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 286 K / Instrument: FEI VITROBOT MARK III / Details: 3s blotting time. |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number real images: 1879 / Average exposure time: 15.0 sec. / Average electron dose: 75.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.8000000000000003 µm / Calibrated defocus min: 1.6 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Details | To build the model for the (CRYZL1)2(PPP1r21)2(GATD1)4 core of the FERRY complex, the obtained crystal structures of CRYZL1 and GATD1 were initially fitted into the corresponding density using the rigid body fitting tool in Chimera. trRosetta, a de novo protein structure prediction algorithm that is based on direct energy minimization with restrained Rosetta, was used to obtain initial models for PPP1r21. The predicted model for the 6-helix bundle domain, containing residues 246 to 498, that matched our experimental density best was subsequently fitted similar as CRYZL1 and GATD1 using rigid body fit. Manual model building for the regions N- and C-terminal 6-helix bundle, which comprise residues 218 to 245 and 499 to 552, respectively, was further guided by secondary structure predictions of individual trRosetta runs for these regions, that include the vertical helix as well as the beginning of the two terminal coiled-coils of PPP1r21. With the resulting combined model, containing residues 2 to 349, 218 to 552 and 8 to 217 of CRYZL1, PPP1r21 and GATD1, respectively, a restrained refinement in PHENIX was performed. In the next step, the model was further refined using a combination of manual building in COOT and real-space refinement in PHENIX. |
|---|---|
| Output model |  PDB-7nd2: |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)