[English] 日本語
 Yorodumi
Yorodumi- EMDB-12263: structure of a POTRA-locked BAM complex in a lateral-open conformation -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12263 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | structure of a POTRA-locked BAM complex in a lateral-open conformation | ||||||||||||||||||||||||
 Map data Map data | Postprocessed masked map of a POTRA-locked BAM complex | ||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationBam protein complex / Gram-negative-bacterium-type cell outer membrane assembly / protein insertion into membrane / cell outer membrane / protein-macromolecule adaptor activity / cell adhesion / response to antibiotic / cell surface / identical protein binding / membrane Similarity search - Function | ||||||||||||||||||||||||
| Biological species |   | ||||||||||||||||||||||||
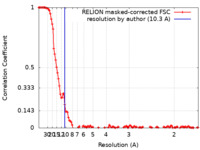
| Method | single particle reconstruction / cryo EM / Resolution: 10.3 Å | ||||||||||||||||||||||||
 Authors Authors | Machin JM / Haysom SF | ||||||||||||||||||||||||
| Funding support |  United Kingdom, United Kingdom,  Belgium, 7 items Belgium, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: The role of membrane destabilisation and protein dynamics in BAM catalysed OMP folding. Authors: Paul White / Samuel F Haysom / Matthew G Iadanza / Anna J Higgins / Jonathan M Machin / James M Whitehouse / Jim E Horne / Bob Schiffrin / Charlotte Carpenter-Platt / Antonio N Calabrese / ...Authors: Paul White / Samuel F Haysom / Matthew G Iadanza / Anna J Higgins / Jonathan M Machin / James M Whitehouse / Jim E Horne / Bob Schiffrin / Charlotte Carpenter-Platt / Antonio N Calabrese / Kelly M Storek / Steven T Rutherford / David J Brockwell / Neil A Ranson / Sheena E Radford /   Abstract: The folding of β-barrel outer membrane proteins (OMPs) in Gram-negative bacteria is catalysed by the β-barrel assembly machinery (BAM). How lateral opening in the β-barrel of the major subunit ...The folding of β-barrel outer membrane proteins (OMPs) in Gram-negative bacteria is catalysed by the β-barrel assembly machinery (BAM). How lateral opening in the β-barrel of the major subunit BamA assists in OMP folding, and the contribution of membrane disruption to BAM catalysis remain unresolved. Here, we use an anti-BamA monoclonal antibody fragment (Fab1) and two disulphide-crosslinked BAM variants (lid-locked (LL), and POTRA-5-locked (P5L)) to dissect these roles. Despite being lethal in vivo, we show that all complexes catalyse folding in vitro, albeit less efficiently than wild-type BAM. CryoEM reveals that while Fab1 and BAM-P5L trap an open-barrel state, BAM-LL contains a mixture of closed and contorted, partially-open structures. Finally, all three complexes globally destabilise the lipid bilayer, while BamA does not, revealing that the BAM lipoproteins are required for this function. Together the results provide insights into the role of BAM structure and lipid dynamics in OMP folding. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12263.map.gz emd_12263.map.gz | 154.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12263-v30.xml emd-12263-v30.xml emd-12263.xml emd-12263.xml | 25.8 KB 25.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12263_fsc.xml emd_12263_fsc.xml | 12.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_12263.png emd_12263.png | 116.6 KB | ||
| Masks |  emd_12263_msk_1.map emd_12263_msk_1.map | 166.4 MB |  Mask map Mask map | |
| Others |  emd_12263_half_map_1.map.gz emd_12263_half_map_1.map.gz emd_12263_half_map_2.map.gz emd_12263_half_map_2.map.gz | 131.5 MB 131.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12263 http://ftp.pdbj.org/pub/emdb/structures/EMD-12263 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12263 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12263 | HTTPS FTP |
-Validation report
| Summary document |  emd_12263_validation.pdf.gz emd_12263_validation.pdf.gz | 469.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_12263_full_validation.pdf.gz emd_12263_full_validation.pdf.gz | 468.6 KB | Display | |
| Data in XML |  emd_12263_validation.xml.gz emd_12263_validation.xml.gz | 19.3 KB | Display | |
| Data in CIF |  emd_12263_validation.cif.gz emd_12263_validation.cif.gz | 25 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12263 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12263 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12263 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12263 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12263.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12263.map.gz / Format: CCP4 / Size: 166.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Postprocessed masked map of a POTRA-locked BAM complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12263_msk_1.map emd_12263_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Halfmap 1 from Refine3D (RELION 3.1)
| File | emd_12263_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Halfmap 1 from Refine3D (RELION 3.1) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Halfmap 2 from Refine3D (RELION 3.1)
| File | emd_12263_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Halfmap 2 from Refine3D (RELION 3.1) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : POTRA-locked beta-barrel assembly machinery (BAM) complex
| Entire | Name: POTRA-locked beta-barrel assembly machinery (BAM) complex |
|---|---|
| Components |
|
-Supramolecule #1: POTRA-locked beta-barrel assembly machinery (BAM) complex
| Supramolecule | Name: POTRA-locked beta-barrel assembly machinery (BAM) complex type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: beta-barrel assembly machinery (BAM) complex (BamABCDE) with POTRA-lock in BamA (G393C G584C) and natural cysteines removed (C690S C700S) |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Molecular weight | Theoretical: 203 KDa |
-Macromolecule #1: Outer membrane protein assembly factor BamA
| Macromolecule | Name: Outer membrane protein assembly factor BamA / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MAMKKLLIAS LLFSSATVYG AEGFVVKDIH FEGLQRVAVG AALLSMPVRT GDTVNDEDIS NTIRALFATG NFEDVRVLRD GDTLLVQVKE RPTIASITFS GNKSVKDDML KQNLEASGVR VGESLDRTTI ADIEKGLEDF YYSVGKYSAS VKAVVTPLPR NRVDLKLVFQ ...String: MAMKKLLIAS LLFSSATVYG AEGFVVKDIH FEGLQRVAVG AALLSMPVRT GDTVNDEDIS NTIRALFATG NFEDVRVLRD GDTLLVQVKE RPTIASITFS GNKSVKDDML KQNLEASGVR VGESLDRTTI ADIEKGLEDF YYSVGKYSAS VKAVVTPLPR NRVDLKLVFQ EGVSAEIQQI NIVGNHAFTT DELISHFQLR DEVPWWNVVG DRKYQKQKLA GDLETLRSYY LDRGYARFNI DSTQVSLTPD KKGIYVTVNI TEGDQYKLSG VEVSGNLAGH SAEIEQLTKI EPGELYNGTK VTKMEDDIKK LLGRYGYAYP RVQSMPEIND ADKTVKLRVN VDAGNRFYVR KIRFEGNDTS KDAVLRREMR QMEGAWLGSD LVDQGKERLN RLGFFETVDT DTQRVPGSPD QVDVVYKVKE RNTGSFNFGI GYGTESGVSF QAGVQQDNWL GTGYAVGING TKNDYQTYAE LSVTNPYFTV DGVSLGGRLF YNDFQADDAD LSDYTNKSYG TDVTLGFPIN EYNSLRAGLG YVHNSLSNMQ PQVAMWRYLY SMGEHPSTSD QDNSFKTDDF TFNYGWTYNK LDRGYFPTDG SRVNLTGKVT IPGSDNEYYK VTLDTATYVP IDDDHKWVVL GRTRWGYGDG LGGKEMPFYE NFYAGGSSTV RGFQSNTIGP KAVYFPHQAS NYDPDYDYEC ATQDGAKDLC KSDDAVGGNA MAVASLEFIT PTPFISDKYA NSVRTSFFWD MGTVWDTNWD SSQYSGYPDY SDPSNIRMSA GIALQWMSPL GPLVFSYAQP FKKYDGDKAE QFQFNIGKTW |
-Macromolecule #2: Outer membrane protein assembly factor BamB
| Macromolecule | Name: Outer membrane protein assembly factor BamB / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MQLRKLLLPG LLSVTLLSGC SLFNSEEDVV KMSPLPTVEN QFTPTTAWST SVGSGIGNFY SNLHPALADN VVYAADRAGL VKALNADDGK EIWSVSLAEK DGWFSKEPAL LSGGVTVSGG HVYIGSEKAQ VYALNTSDGT VAWQTKVAGE ALSRPVVSDG LVLIHTSNGQ ...String: MQLRKLLLPG LLSVTLLSGC SLFNSEEDVV KMSPLPTVEN QFTPTTAWST SVGSGIGNFY SNLHPALADN VVYAADRAGL VKALNADDGK EIWSVSLAEK DGWFSKEPAL LSGGVTVSGG HVYIGSEKAQ VYALNTSDGT VAWQTKVAGE ALSRPVVSDG LVLIHTSNGQ LQALNEADGA VKWTVNLDMP SLSLRGESAP TTAFGAAVVG GDNGRVSAVL MEQGQMIWQQ RISQATGSTE IDRLSDVDTT PVVVNGVVFA LAYNGNLTAL DLRSGQIMWK RELGSVNDFI VDGNRIYLVD QNDRVMALTI DGGVTLWTQS DLLHRLLTSP VLYNGNLVVG DSEGYLHWIN VEDGRFVAQQ KVDSSGFQTE PVAADGKLLI QAKDGTVYSI TR |
-Macromolecule #3: Outer membrane protein assembly factor BamC
| Macromolecule | Name: Outer membrane protein assembly factor BamC / type: protein_or_peptide / ID: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MAYSVQKSRL AKVAGVSLVL LLAACSSDSR YKRQVSGDEA YLEAAPLAEL HAPAGMILPV TSGDYAIPVT NGSGAVGKAL DIRPPAQPLA LVSGARTQFT GDTASLLVEN GRGNTLWPQV VSVLQAKNYT ITQRDDAGQT LTTDWVQWNR LDEDEQYRGR YQISVKPQGY ...String: MAYSVQKSRL AKVAGVSLVL LLAACSSDSR YKRQVSGDEA YLEAAPLAEL HAPAGMILPV TSGDYAIPVT NGSGAVGKAL DIRPPAQPLA LVSGARTQFT GDTASLLVEN GRGNTLWPQV VSVLQAKNYT ITQRDDAGQT LTTDWVQWNR LDEDEQYRGR YQISVKPQGY QQAVTVKLLN LEQAGKPVAD AASMQRYSTE MMNVISAGLD KSATDAANAA QNRASTTMDV QSAADDTGLP MLVVRGPFNV VWQRLPAALE KVGMKVTDST RSQGNMAVTY KPLSDSDWQE LGASDPGLAS GDYKLQVGDL DNRSSLQFID PKGHTLTQSQ NDALVAVFQA AFSK |
-Macromolecule #4: Outer membrane protein assembly factor BamD
| Macromolecule | Name: Outer membrane protein assembly factor BamD / type: protein_or_peptide / ID: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MTRMKYLVAA ATLSLFLAGC SGSKEEVPDN PPNEIYATAQ QKLQDGNWRQ AITQLEALDN RYPFGPYSQQ VQLDLIYAYY KNADLPLAQA AIDRFIRLNP THPNIDYVMY MRGLTNMALD DSALQGFFGV DRSDRDPQHA RAAFSDFSKL VRGYPNSQYT TDATKRLVFL ...String: MTRMKYLVAA ATLSLFLAGC SGSKEEVPDN PPNEIYATAQ QKLQDGNWRQ AITQLEALDN RYPFGPYSQQ VQLDLIYAYY KNADLPLAQA AIDRFIRLNP THPNIDYVMY MRGLTNMALD DSALQGFFGV DRSDRDPQHA RAAFSDFSKL VRGYPNSQYT TDATKRLVFL KDRLAKYEYS VAEYYTERGA WVAVVNRVEG MLRDYPDTQA TRDALPLMEN AYRQMQMNAQ AEKVAKIIAA NSSNT |
-Macromolecule #5: Outer membrane protein assembly factor BamE
| Macromolecule | Name: Outer membrane protein assembly factor BamE / type: protein_or_peptide / ID: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Sequence | String: MRCKTLTAAA AVLLMLTAGC STLERVVYRP DINQGNYLTA NDVSKIRVGM TQQQVAYALG TPLMSDPFGT NTWFYVFRQQ PGHEGVTQQT LTLTFNSSGV LTNIDNKPAL SGN |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2.3 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| ||||||||||||
| Grid | Model: Quantifoil R0.6/1 / Material: COPPER / Mesh: 400 / Pretreatment - Type: GLOW DISCHARGE / Details: Glow discharge using GloqQube Plus, 30s, 60 mA | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy #1
Electron microscopy #1
| Microscopy ID | 1 |
|---|---|
| Microscope | FEI TITAN KRIOS |
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Image recording ID: 1 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Electron microscopy #1~
Electron microscopy #1~
| Microscopy ID | 1 |
|---|---|
| Microscope | FEI TITAN KRIOS |
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Image recording ID: 2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 58.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 165000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller

























 Z
Z Y
Y X
X