+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8gj3 | ||||||
|---|---|---|---|---|---|---|---|
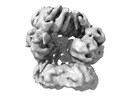
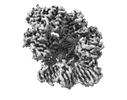
| Title | E. coli clamp loader on primed template DNA | ||||||
 Components Components |
| ||||||
 Keywords Keywords | DNA BINDING PROTEIN/DNA / clamp loader /  DNA clamp / DNA clamp /  AAA+ / AAA+ /  ATPase / DNA BINDING PROTEIN-DNA complex ATPase / DNA BINDING PROTEIN-DNA complex | ||||||
| Function / homology |  Function and homology information Function and homology information DNA polymerase III, clamp loader complex / DNA polymerase III, clamp loader complex /  DNA polymerase III complex / DNA polymerase III complex /  replisome / DNA polymerase processivity factor activity / 3'-5' exonuclease activity / ribonucleoside triphosphate phosphatase activity / response to radiation / DNA-templated DNA replication / replisome / DNA polymerase processivity factor activity / 3'-5' exonuclease activity / ribonucleoside triphosphate phosphatase activity / response to radiation / DNA-templated DNA replication /  DNA replication / DNA replication /  DNA-directed DNA polymerase ... DNA-directed DNA polymerase ... DNA polymerase III, clamp loader complex / DNA polymerase III, clamp loader complex /  DNA polymerase III complex / DNA polymerase III complex /  replisome / DNA polymerase processivity factor activity / 3'-5' exonuclease activity / ribonucleoside triphosphate phosphatase activity / response to radiation / DNA-templated DNA replication / replisome / DNA polymerase processivity factor activity / 3'-5' exonuclease activity / ribonucleoside triphosphate phosphatase activity / response to radiation / DNA-templated DNA replication /  DNA replication / DNA replication /  DNA-directed DNA polymerase / DNA-directed DNA polymerase /  DNA-directed DNA polymerase activity / DNA-directed DNA polymerase activity /  ATP hydrolysis activity / ATP hydrolysis activity /  DNA binding / DNA binding /  ATP binding / identical protein binding ATP binding / identical protein bindingSimilarity search - Function | ||||||
| Biological species |   Escherichia coli K-12 (bacteria) Escherichia coli K-12 (bacteria)  Escherichia coli (E. coli) Escherichia coli (E. coli) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.8 Å cryo EM / Resolution: 2.8 Å | ||||||
 Authors Authors | Oakley, A.J. / Xu, Z.-Q. / Dixon, N.E. | ||||||
| Funding support |  Australia, 1items Australia, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Structural characterisation of the complete cycle of sliding clamp loading in E. coli Authors: Xu, Z.-Q. / Oakley, A.J. / Dixon, N.E. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8gj3.cif.gz 8gj3.cif.gz | 890.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8gj3.ent.gz pdb8gj3.ent.gz | 582.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8gj3.json.gz 8gj3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gj/8gj3 https://data.pdbj.org/pub/pdb/validation_reports/gj/8gj3 ftp://data.pdbj.org/pub/pdb/validation_reports/gj/8gj3 ftp://data.pdbj.org/pub/pdb/validation_reports/gj/8gj3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  40084MC  8giyC  8gizC  8gj0C  8gj1C  8gj2C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-DNA polymerase III subunit ... , 4 types, 6 molecules ABCDEF
| #1: Protein |  DNA polymerase III holoenzyme DNA polymerase III holoenzymeMass: 38745.574 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli K-12 (bacteria) / Gene: holA, b0640, JW0635 / Production host: Escherichia coli K-12 (bacteria) / Gene: holA, b0640, JW0635 / Production host:   Escherichia coli BL21 (bacteria) / References: UniProt: P28630, Escherichia coli BL21 (bacteria) / References: UniProt: P28630,  DNA-directed DNA polymerase DNA-directed DNA polymerase | ||||
|---|---|---|---|---|---|
| #2: Protein |  DNA polymerase III holoenzyme / DNA polymerase III subunit gamma DNA polymerase III holoenzyme / DNA polymerase III subunit gammaMass: 71228.648 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli K-12 (bacteria) / Gene: dnaX, dnaZ, dnaZX, b0470, JW0459 / Production host: Escherichia coli K-12 (bacteria) / Gene: dnaX, dnaZ, dnaZX, b0470, JW0459 / Production host:   Escherichia coli BL21 (bacteria) / References: UniProt: P06710, Escherichia coli BL21 (bacteria) / References: UniProt: P06710,  DNA-directed DNA polymerase DNA-directed DNA polymerase#3: Protein | |  DNA polymerase III holoenzyme DNA polymerase III holoenzymeMass: 36980.484 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli K-12 (bacteria) / Gene: holB, b1099, JW1085 / Production host: Escherichia coli K-12 (bacteria) / Gene: holB, b1099, JW1085 / Production host:   Escherichia coli BL21 (bacteria) / References: UniProt: P28631, Escherichia coli BL21 (bacteria) / References: UniProt: P28631,  DNA-directed DNA polymerase DNA-directed DNA polymerase#4: Protein | |  DNA polymerase III holoenzyme DNA polymerase III holoenzymeMass: 15188.276 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli K-12 (bacteria) / Gene: holD, b4372, JW4334 / Production host: Escherichia coli K-12 (bacteria) / Gene: holD, b4372, JW4334 / Production host:   Escherichia coli BL21 (bacteria) / References: UniProt: P28632, Escherichia coli BL21 (bacteria) / References: UniProt: P28632,  DNA-directed DNA polymerase DNA-directed DNA polymerase |
-DNA chain , 2 types, 2 molecules XY
| #5: DNA chain | Mass: 4856.191 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)   Escherichia coli (E. coli) Escherichia coli (E. coli) |
|---|---|
| #6: DNA chain | Mass: 7969.154 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)   Escherichia coli (E. coli) Escherichia coli (E. coli) |
-Non-polymers , 4 types, 13 molecules 






| #7: Chemical |  Adenosine diphosphate Adenosine diphosphate#8: Chemical | #9: Chemical | #10: Chemical | ChemComp-ZN / |
|---|
-Details
| Has ligand of interest | N |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: E. coli clamp loader on primed template DNA / Type: COMPLEX Details: Clamp loader complex composed of DNA polymerase III delta tau(3) delta' chi and psi subunits. Clamp composed of DNA polymerase III beta(2) subunits. Entity ID: #1-#6 / Source: MULTIPLE SOURCES | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.333 MDa / Experimental value: NO | |||||||||||||||||||||||||||||||||||||||||||||
| Source (natural) | Organism:   Escherichia coli (E. coli) / Strain: K-12 Escherichia coli (E. coli) / Strain: K-12 | |||||||||||||||||||||||||||||||||||||||||||||
| Source (recombinant) | Organism:   Escherichia coli (E. coli) / Strain: BL21 Escherichia coli (E. coli) / Strain: BL21 | |||||||||||||||||||||||||||||||||||||||||||||
| Buffer solution | pH: 7.6 Details: 30 mM Tris-HCl pH 7.6, 5 mM MgCl2, 2 mM ADP, 0.5 mM AlCl3, 5 mM NaF, 5 mM dithiothreitol, 0.25 mM EDTA, 2% glycerol. | |||||||||||||||||||||||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YESDetails: 30 microL of 6.0 microM delta/tau3/delta' complex was mixed with chi/psi complex, beta, and p/t DNA (annealed DNA oligonucleotides) at a molar ratio of 1:1.3 and dialysed at 4 degrees C ...Details: 30 microL of 6.0 microM delta/tau3/delta' complex was mixed with chi/psi complex, beta, and p/t DNA (annealed DNA oligonucleotides) at a molar ratio of 1:1.3 and dialysed at 4 degrees C against 250 mL of 30 mM Tris-HCl pH 7.6, 5 mM MgCl2, 2 mM ADP, 0.5 mM AlCl3, 5 mM NaF, 5 mM dithiothreitol, 0.25 mM EDTA, 2% glycerol. | |||||||||||||||||||||||||||||||||||||||||||||
| Specimen support | Details: Used a Zepto Low-pressure plasma systems (PLASMA CLEANER) (Diener Electronic) at 10% power for 120 seconds. Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: UltrAuFoil R1.2/1.3 | |||||||||||||||||||||||||||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 279 K Details: 3 uL of sample was applied onto a Ultrafoil Au R1.2/1.3 grid. Blot for 4.5 s with no extra force before plunging into liquid ethane. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1300 nm / Nominal defocus min: 400 nm / Alignment procedure: COMA FREE Bright-field microscopy / Nominal defocus max: 1300 nm / Nominal defocus min: 400 nm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 6 sec. / Electron dose: 50 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 2909 |
| EM imaging optics | Energyfilter name : GIF Bioquantum / Details: unfiltered mode : GIF Bioquantum / Details: unfiltered mode |
| Image scans | Width: 4096 / Height: 4096 / Movie frames/image: 60 / Used frames/image: 1-60 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||
CTF correction | Details: CTF refinement per-particle was performed in RELION before final 3D reconstruction. Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 645000 | ||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 308000 / Algorithm: BACK PROJECTION / Num. of class averages: 2 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building |
| ||||||||||||||||||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 124.83 Å2 | ||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






 PDBj
PDBj

















































