+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8dvr | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
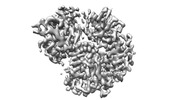
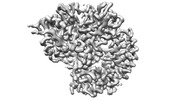
| Title | Cryo-EM structure of RIG-I bound to the end of p3SLR30 (+AMPPNP) | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | Hydrolase/RNA /  ribonucleoprotein complex / RNA sensor / RIG-I like receptor / Hydrolase-RNA complex ribonucleoprotein complex / RNA sensor / RIG-I like receptor / Hydrolase-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of type III interferon production / RIG-I signaling pathway / positive regulation of myeloid dendritic cell cytokine production / OAS antiviral response / detection of virus / NF-kB activation through FADD/RIP-1 pathway mediated by caspase-8 and -10 / positive regulation of response to cytokine stimulus / positive regulation of granulocyte macrophage colony-stimulating factor production /  pattern recognition receptor activity / TRAF6 mediated IRF7 activation ...regulation of type III interferon production / RIG-I signaling pathway / positive regulation of myeloid dendritic cell cytokine production / OAS antiviral response / detection of virus / NF-kB activation through FADD/RIP-1 pathway mediated by caspase-8 and -10 / positive regulation of response to cytokine stimulus / positive regulation of granulocyte macrophage colony-stimulating factor production / pattern recognition receptor activity / TRAF6 mediated IRF7 activation ...regulation of type III interferon production / RIG-I signaling pathway / positive regulation of myeloid dendritic cell cytokine production / OAS antiviral response / detection of virus / NF-kB activation through FADD/RIP-1 pathway mediated by caspase-8 and -10 / positive regulation of response to cytokine stimulus / positive regulation of granulocyte macrophage colony-stimulating factor production /  pattern recognition receptor activity / TRAF6 mediated IRF7 activation / cellular response to exogenous dsRNA / response to exogenous dsRNA / cytoplasmic pattern recognition receptor signaling pathway / positive regulation of interferon-alpha production / antiviral innate immune response / TRAF6 mediated NF-kB activation / bicellular tight junction / pattern recognition receptor activity / TRAF6 mediated IRF7 activation / cellular response to exogenous dsRNA / response to exogenous dsRNA / cytoplasmic pattern recognition receptor signaling pathway / positive regulation of interferon-alpha production / antiviral innate immune response / TRAF6 mediated NF-kB activation / bicellular tight junction /  regulation of cell migration / positive regulation of defense response to virus by host / positive regulation of interferon-beta production / Negative regulators of DDX58/IFIH1 signaling / positive regulation of interleukin-8 production / response to virus / DDX58/IFIH1-mediated induction of interferon-alpha/beta / ISG15 antiviral mechanism / ruffle membrane / positive regulation of interleukin-6 production / SARS-CoV-1 activates/modulates innate immune responses / positive regulation of DNA-binding transcription factor activity / Ovarian tumor domain proteases / regulation of cell migration / positive regulation of defense response to virus by host / positive regulation of interferon-beta production / Negative regulators of DDX58/IFIH1 signaling / positive regulation of interleukin-8 production / response to virus / DDX58/IFIH1-mediated induction of interferon-alpha/beta / ISG15 antiviral mechanism / ruffle membrane / positive regulation of interleukin-6 production / SARS-CoV-1 activates/modulates innate immune responses / positive regulation of DNA-binding transcription factor activity / Ovarian tumor domain proteases /  actin cytoskeleton / positive regulation of tumor necrosis factor production / actin cytoskeleton / positive regulation of tumor necrosis factor production /  double-stranded RNA binding / double-stranded RNA binding /  gene expression / TRAF3-dependent IRF activation pathway / gene expression / TRAF3-dependent IRF activation pathway /  double-stranded DNA binding / defense response to virus / double-stranded DNA binding / defense response to virus /  RNA helicase activity / RNA helicase activity /  single-stranded RNA binding / Ub-specific processing proteases / single-stranded RNA binding / Ub-specific processing proteases /  RNA helicase / RNA helicase /  ribonucleoprotein complex / ribonucleoprotein complex /  innate immune response / innate immune response /  ubiquitin protein ligase binding / positive regulation of gene expression / GTP binding / SARS-CoV-2 activates/modulates innate and adaptive immune responses / ubiquitin protein ligase binding / positive regulation of gene expression / GTP binding / SARS-CoV-2 activates/modulates innate and adaptive immune responses /  ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / zinc ion binding / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / zinc ion binding /  ATP binding / identical protein binding / ATP binding / identical protein binding /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.3 Å cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Wang, W. / Pyle, A.M. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2022 Journal: Mol Cell / Year: 2022Title: The RIG-I receptor adopts two different conformations for distinguishing host from viral RNA ligands. Authors: Wenshuai Wang / Anna Marie Pyle /  Abstract: RIG-I is an essential innate immune receptor for detecting and responding to infection by RNA viruses. RIG-I specifically recognizes the unique molecular features of viral RNA molecules and ...RIG-I is an essential innate immune receptor for detecting and responding to infection by RNA viruses. RIG-I specifically recognizes the unique molecular features of viral RNA molecules and selectively distinguishes them from closely related RNAs abundant in host cells. The physical basis for this exquisite selectivity is revealed through a series of high-resolution cryo-EM structures of RIG-I in complex with host and viral RNA ligands. These studies demonstrate that RIG-I actively samples double-stranded RNAs in the cytoplasm and distinguishes them by adopting two different types of protein folds. Upon binding viral RNA, RIG-I adopts a high-affinity conformation that is conducive to signaling, while host RNA induces an autoinhibited conformation that stimulates RNA release. By coupling protein folding with RNA binding selectivity, RIG-I distinguishes RNA molecules that differ by as little as one phosphate group, thereby explaining the molecular basis for selective antiviral sensing and the induction of autoimmunity upon RIG-I dysregulation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8dvr.cif.gz 8dvr.cif.gz | 139.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8dvr.ent.gz pdb8dvr.ent.gz | 104.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8dvr.json.gz 8dvr.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dv/8dvr https://data.pdbj.org/pub/pdb/validation_reports/dv/8dvr ftp://data.pdbj.org/pub/pdb/validation_reports/dv/8dvr ftp://data.pdbj.org/pub/pdb/validation_reports/dv/8dvr | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  27743MC  7tnxC  7tnyC  7tnzC  7to0C  7to1C  7to2C  8dvsC  8dvuC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 106740.555 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: DDX58 / Production host: Homo sapiens (human) / Gene: DDX58 / Production host:   Escherichia coli (E. coli) / References: UniProt: O95786, Escherichia coli (E. coli) / References: UniProt: O95786,  RNA helicase RNA helicase |
|---|---|
| #2: RNA chain | Mass: 19513.580 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)   Homo sapiens (human) Homo sapiens (human) |
| #3: Chemical | ChemComp-ZN / |
| #4: Chemical | ChemComp-GTP /  Guanosine triphosphate Guanosine triphosphate |
| Has ligand of interest | Y |
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Complex of RIG-I with p3SLR30 with AMPPNP addition / Type: COMPLEX / Entity ID: #2 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 3000 nm / Nominal defocus min: 1200 nm Bright-field microscopy / Nominal defocus max: 3000 nm / Nominal defocus min: 1200 nm |
| Image recording | Electron dose: 60 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.17.1_3660: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Type: NONE | ||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||
3D reconstruction | Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 692833 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | B value: 157.95 | ||||||||||||||||||||||||
| Atomic model building | PDB-ID: 7TO1 | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj










































