+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6qw3 | ||||||
|---|---|---|---|---|---|---|---|
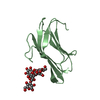
| Title | Calcium-bound gelsolin domain 2 | ||||||
 Components Components | Gelsolin | ||||||
 Keywords Keywords |  STRUCTURAL PROTEIN / STRUCTURAL PROTEIN /  amyloidosis / amyloidosis /  gelsolin / actin-binding gelsolin / actin-binding | ||||||
| Function / homology |  Function and homology information Function and homology informationstriated muscle atrophy / regulation of establishment of T cell polarity / regulation of plasma membrane raft polarization / regulation of receptor clustering /  renal protein absorption / positive regulation of keratinocyte apoptotic process / positive regulation of protein processing in phagocytic vesicle / positive regulation of actin nucleation / phosphatidylinositol 3-kinase catalytic subunit binding / positive regulation of cysteine-type endopeptidase activity involved in apoptotic signaling pathway ...striated muscle atrophy / regulation of establishment of T cell polarity / regulation of plasma membrane raft polarization / regulation of receptor clustering / renal protein absorption / positive regulation of keratinocyte apoptotic process / positive regulation of protein processing in phagocytic vesicle / positive regulation of actin nucleation / phosphatidylinositol 3-kinase catalytic subunit binding / positive regulation of cysteine-type endopeptidase activity involved in apoptotic signaling pathway ...striated muscle atrophy / regulation of establishment of T cell polarity / regulation of plasma membrane raft polarization / regulation of receptor clustering /  renal protein absorption / positive regulation of keratinocyte apoptotic process / positive regulation of protein processing in phagocytic vesicle / positive regulation of actin nucleation / phosphatidylinositol 3-kinase catalytic subunit binding / positive regulation of cysteine-type endopeptidase activity involved in apoptotic signaling pathway / actin cap / sequestering of actin monomers / renal protein absorption / positive regulation of keratinocyte apoptotic process / positive regulation of protein processing in phagocytic vesicle / positive regulation of actin nucleation / phosphatidylinositol 3-kinase catalytic subunit binding / positive regulation of cysteine-type endopeptidase activity involved in apoptotic signaling pathway / actin cap / sequestering of actin monomers /  regulation of podosome assembly / regulation of podosome assembly /  myosin II binding / negative regulation of viral entry into host cell / actin filament severing / actin filament capping / barbed-end actin filament capping / actin polymerization or depolymerization / actin filament depolymerization / cell projection assembly / cardiac muscle cell contraction / myosin II binding / negative regulation of viral entry into host cell / actin filament severing / actin filament capping / barbed-end actin filament capping / actin polymerization or depolymerization / actin filament depolymerization / cell projection assembly / cardiac muscle cell contraction /  podosome / Sensory processing of sound by outer hair cells of the cochlea / relaxation of cardiac muscle / podosome / Sensory processing of sound by outer hair cells of the cochlea / relaxation of cardiac muscle /  phagocytosis, engulfment / cortical actin cytoskeleton / hepatocyte apoptotic process / phagocytosis, engulfment / cortical actin cytoskeleton / hepatocyte apoptotic process /  cilium assembly / cilium assembly /  sarcoplasm / Caspase-mediated cleavage of cytoskeletal proteins / phagocytic vesicle / sarcoplasm / Caspase-mediated cleavage of cytoskeletal proteins / phagocytic vesicle /  phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / response to muscle stretch / phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / response to muscle stretch /  central nervous system development / actin filament organization / protein destabilization / cellular response to type II interferon / central nervous system development / actin filament organization / protein destabilization / cellular response to type II interferon /  actin filament binding / actin filament binding /  actin cytoskeleton / actin cytoskeleton /  lamellipodium / lamellipodium /  actin binding / secretory granule lumen / blood microparticle / ficolin-1-rich granule lumen / amyloid fibril formation / Amyloid fiber formation / actin binding / secretory granule lumen / blood microparticle / ficolin-1-rich granule lumen / amyloid fibril formation / Amyloid fiber formation /  focal adhesion / focal adhesion /  calcium ion binding / Neutrophil degranulation / positive regulation of gene expression / calcium ion binding / Neutrophil degranulation / positive regulation of gene expression /  extracellular space / extracellular exosome / extracellular region / extracellular space / extracellular exosome / extracellular region /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3 Å MOLECULAR REPLACEMENT / Resolution: 1.3 Å | ||||||
 Authors Authors | Scalone, E. / Boni, F. / Milani, M. / Mastrangelo, E. / de Rosa, M. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Biochem.Biophys.Res.Commun. / Year: 2019 Journal: Biochem.Biophys.Res.Commun. / Year: 2019Title: High-resolution crystal structure of gelsolin domain 2 in complex with the physiological calcium ion. Authors: Bollati, M. / Scalone, E. / Boni, F. / Mastrangelo, E. / Giorgino, T. / Milani, M. / de Rosa, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6qw3.cif.gz 6qw3.cif.gz | 66.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6qw3.ent.gz pdb6qw3.ent.gz | 47.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6qw3.json.gz 6qw3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qw/6qw3 https://data.pdbj.org/pub/pdb/validation_reports/qw/6qw3 ftp://data.pdbj.org/pub/pdb/validation_reports/qw/6qw3 ftp://data.pdbj.org/pub/pdb/validation_reports/qw/6qw3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1kcqS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
| ||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein |  / AGEL / Actin-depolymerizing factor / ADF / Brevin / AGEL / Actin-depolymerizing factor / ADF / BrevinMass: 13166.648 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: gelsolin domain 2 (G2) / Source: (gene. exp.)   Homo sapiens (human) / Gene: GSN / Production host: Homo sapiens (human) / Gene: GSN / Production host:   Escherichia coli (E. coli) / References: UniProt: P06396 Escherichia coli (E. coli) / References: UniProt: P06396 |
|---|---|
| #2: Chemical | ChemComp-CA / |
| #3: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.8 Å3/Da / Density % sol: 31.74 % |
|---|---|
Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 5.5 / Details: 36% PEG5000 MME, 0.1M Sodium Acetate |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I04 / Wavelength: 0.9795 Å / Beamline: I04 / Wavelength: 0.9795 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Feb 10, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9795 Å / Relative weight: 1 : 0.9795 Å / Relative weight: 1 |
| Reflection | Resolution: 1.3→70.82 Å / Num. obs: 24096 / % possible obs: 99.5 % / Redundancy: 10.8 % / CC1/2: 0.999 / Rmerge(I) obs: 0.077 / Rpim(I) all: 0.023 / Rrim(I) all: 0.08 / Net I/σ(I): 14 |
| Reflection shell | Resolution: 1.3→1.32 Å / Redundancy: 4.3 % / Rmerge(I) obs: 0.764 / Mean I/σ(I) obs: 1.8 / Num. unique obs: 1103 / CC1/2: 0.579 / Rpim(I) all: 0.409 / Rrim(I) all: 0.874 / % possible all: 92.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1KCQ Resolution: 1.3→40.995 Å / SU ML: 0.11 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 17.75
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.3→40.995 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




