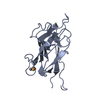
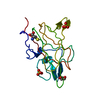
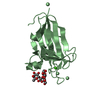
Entry Database : PDB / ID : 4v11Title Structure of Synaptotagmin-1 with SV2A peptide phosphorylated at Thr84 SYNAPTIC VESICLE GLYCOPROTEIN 2A SYNAPTOTAGMIN-1 Keywords Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species HOMO SAPIENS (human)Method / / Resolution : 1.95 Å Authors Zhang, N. / Gordon, S.L. / Fritsch, M.J. / Esoof, N. / Campbell, D. / Gourlay, R. / Velupillai, S. / Macartney, T. / Peggie, M. / vanAalten, D.M.F. ...Zhang, N. / Gordon, S.L. / Fritsch, M.J. / Esoof, N. / Campbell, D. / Gourlay, R. / Velupillai, S. / Macartney, T. / Peggie, M. / vanAalten, D.M.F. / Cousin, M.A. / Alessi, D.R. Journal : J.Neurosci. / Year : 2015Title : Phosphorylation of Synaptic Vesicle Protein 2A at Thr84 by Casein Kinase 1 Family Kinases Controls the Specific Retrieval of Synaptotagmin-1.Authors : Zhang, N. / Gordon, S.L. / Fritsch, M.J. / Esoof, N. / Campbell, D.G. / Gourlay, R. / Velupillai, S. / Macartney, T. / Peggie, M. / Van Aalten, D.M.F. / Cousin, M.A. / Alessi, D.R. History Deposition Sep 22, 2014 Deposition site / Processing site Revision 1.0 Feb 25, 2015 Provider / Type Revision 1.1 Jun 28, 2017 Group / Category / Item Revision 1.2 Jan 10, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Other / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_database_status / pdbx_initial_refinement_model / pdbx_struct_conn_angle / struct_conn / struct_conn_type / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_sf / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr2_auth_seq_id / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn_type.id / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords SIGNALING PROTEIN
SIGNALING PROTEIN Function and homology information
Function and homology information syntaxin-3 binding / calcium-dependent activation of synaptic vesicle fusion / Acetylcholine Neurotransmitter Release Cycle / Toxicity of botulinum toxin type A (botA) / regulation of regulated secretory pathway / calcium ion sensor activity / clathrin-sculpted monoamine transport vesicle membrane / Toxicity of botulinum toxin type B (botB) / spontaneous neurotransmitter secretion / clathrin-sculpted gamma-aminobutyric acid transport vesicle membrane / Serotonin Neurotransmitter Release Cycle / dense core granule / chromaffin granule membrane / GABA synthesis, release, reuptake and degradation / calcium ion-regulated exocytosis of neurotransmitter / Dopamine Neurotransmitter Release Cycle / Norepinephrine Neurotransmitter Release Cycle / vesicle docking / regulation of calcium ion-dependent exocytosis / exocytic vesicle / positive regulation of dopamine secretion / protein heterooligomerization / Glutamate Neurotransmitter Release Cycle /
syntaxin-3 binding / calcium-dependent activation of synaptic vesicle fusion / Acetylcholine Neurotransmitter Release Cycle / Toxicity of botulinum toxin type A (botA) / regulation of regulated secretory pathway / calcium ion sensor activity / clathrin-sculpted monoamine transport vesicle membrane / Toxicity of botulinum toxin type B (botB) / spontaneous neurotransmitter secretion / clathrin-sculpted gamma-aminobutyric acid transport vesicle membrane / Serotonin Neurotransmitter Release Cycle / dense core granule / chromaffin granule membrane / GABA synthesis, release, reuptake and degradation / calcium ion-regulated exocytosis of neurotransmitter / Dopamine Neurotransmitter Release Cycle / Norepinephrine Neurotransmitter Release Cycle / vesicle docking / regulation of calcium ion-dependent exocytosis / exocytic vesicle / positive regulation of dopamine secretion / protein heterooligomerization / Glutamate Neurotransmitter Release Cycle /  regulation of exocytosis / neurotransmitter secretion / positive regulation of dendrite extension / calcium-dependent phospholipid binding / neuron projection terminus / Neurexins and neuroligins /
regulation of exocytosis / neurotransmitter secretion / positive regulation of dendrite extension / calcium-dependent phospholipid binding / neuron projection terminus / Neurexins and neuroligins /  syntaxin-1 binding /
syntaxin-1 binding /  syntaxin binding / synaptic vesicle priming /
syntaxin binding / synaptic vesicle priming /  clathrin binding / low-density lipoprotein particle receptor binding /
clathrin binding / low-density lipoprotein particle receptor binding /  phosphatidylserine binding / regulation of dopamine secretion / presynaptic active zone /
phosphatidylserine binding / regulation of dopamine secretion / presynaptic active zone /  excitatory synapse / transmembrane transporter activity / synaptic vesicle endocytosis / detection of calcium ion / GABA-ergic synapse / positive regulation of synaptic transmission /
excitatory synapse / transmembrane transporter activity / synaptic vesicle endocytosis / detection of calcium ion / GABA-ergic synapse / positive regulation of synaptic transmission /  regulation of synaptic transmission, glutamatergic /
regulation of synaptic transmission, glutamatergic /  phosphatidylinositol-4,5-bisphosphate binding / cellular response to calcium ion / hippocampal mossy fiber to CA3 synapse /
phosphatidylinositol-4,5-bisphosphate binding / cellular response to calcium ion / hippocampal mossy fiber to CA3 synapse /  SNARE binding / clathrin-coated endocytic vesicle membrane /
SNARE binding / clathrin-coated endocytic vesicle membrane /  neuromuscular junction / synaptic vesicle membrane / intracellular calcium ion homeostasis / calcium-dependent protein binding / cell-cell junction /
neuromuscular junction / synaptic vesicle membrane / intracellular calcium ion homeostasis / calcium-dependent protein binding / cell-cell junction /  synaptic vesicle / Cargo recognition for clathrin-mediated endocytosis /
synaptic vesicle / Cargo recognition for clathrin-mediated endocytosis /  presynaptic membrane /
presynaptic membrane /  Clathrin-mediated endocytosis / chemical synaptic transmission /
Clathrin-mediated endocytosis / chemical synaptic transmission /  cell differentiation /
cell differentiation /  calmodulin binding / neuron projection / protein heterodimerization activity /
calmodulin binding / neuron projection / protein heterodimerization activity /  axon / neuronal cell body / glutamatergic synapse /
axon / neuronal cell body / glutamatergic synapse /  dendrite /
dendrite /  calcium ion binding /
calcium ion binding /  protein kinase binding /
protein kinase binding /  Golgi apparatus /
Golgi apparatus /  endoplasmic reticulum / identical protein binding /
endoplasmic reticulum / identical protein binding /  plasma membrane /
plasma membrane /  cytoplasm
cytoplasm
 HOMO SAPIENS (human)
HOMO SAPIENS (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.95 Å
MOLECULAR REPLACEMENT / Resolution: 1.95 Å  Authors
Authors Citation
Citation Journal: J.Neurosci. / Year: 2015
Journal: J.Neurosci. / Year: 2015 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4v11.cif.gz
4v11.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4v11.ent.gz
pdb4v11.ent.gz PDB format
PDB format 4v11.json.gz
4v11.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/v1/4v11
https://data.pdbj.org/pub/pdb/validation_reports/v1/4v11 ftp://data.pdbj.org/pub/pdb/validation_reports/v1/4v11
ftp://data.pdbj.org/pub/pdb/validation_reports/v1/4v11
 Links
Links Assembly
Assembly
 Components
Components / SYNAPTOTAGMIN I / SYTI / P65
/ SYNAPTOTAGMIN I / SYTI / P65
 HOMO SAPIENS (human) / Plasmid: PGEX6P-1 TEVB SYT1 270-END / Production host:
HOMO SAPIENS (human) / Plasmid: PGEX6P-1 TEVB SYT1 270-END / Production host: 
 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P21579
ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P21579
 HOMO SAPIENS (human) / References: UniProt: Q7L0J3
HOMO SAPIENS (human) / References: UniProt: Q7L0J3 Glycerol
Glycerol Water
Water X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation
 ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178
ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178  : 1.54178 Å / Relative weight: 1
: 1.54178 Å / Relative weight: 1  Processing
Processing :
:  MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller









 PDBj
PDBj












