[English] 日本語
 Yorodumi
Yorodumi- PDB-4ahl: P112L - Angiogenin mutants and amyotrophic lateral sclerosis - a ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4ahl | ||||||
|---|---|---|---|---|---|---|---|
| Title | P112L - Angiogenin mutants and amyotrophic lateral sclerosis - a biochemical and biological analysis | ||||||
 Components Components | ANGIOGENIN | ||||||
 Keywords Keywords | HYDROLASE / NEOVASCULARISATION | ||||||
| Function / homology |  Function and homology information Function and homology informationactivation of phospholipase A2 activity / angiogenin-PRI complex / diacylglycerol biosynthetic process / tRNA-derived small RNA (tsRNA or tRNA-related fragment, tRF) biogenesis / tRNA decay / cell communication / Hydrolases; Acting on ester bonds; Endoribonucleases producing 3'-phosphomonoesters / Adherens junctions interactions / : / oocyte maturation ...activation of phospholipase A2 activity / angiogenin-PRI complex / diacylglycerol biosynthetic process / tRNA-derived small RNA (tsRNA or tRNA-related fragment, tRF) biogenesis / tRNA decay / cell communication / Hydrolases; Acting on ester bonds; Endoribonucleases producing 3'-phosphomonoesters / Adherens junctions interactions / : / oocyte maturation / homeostatic process / positive regulation of phosphorylation / rRNA transcription / basement membrane / RNA nuclease activity / ovarian follicle development / positive regulation of endothelial cell proliferation / RNA endonuclease activity / actin filament polymerization / activation of protein kinase B activity / negative regulation of smooth muscle cell proliferation / response to hormone / peptide binding / placenta development / positive regulation of protein secretion / chromosome / antimicrobial humoral immune response mediated by antimicrobial peptide / actin cytoskeleton / cell migration / heparin binding / actin binding / antibacterial humoral response / growth cone / cytoplasmic vesicle / angiogenesis / endonuclease activity / response to hypoxia / rRNA binding / negative regulation of translation / defense response to Gram-positive bacterium / copper ion binding / innate immune response / signaling receptor binding / neuronal cell body / nucleolus / protein homodimerization activity / DNA binding / extracellular space / extracellular region / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.053 Å MOLECULAR REPLACEMENT / Resolution: 2.053 Å | ||||||
 Authors Authors | Thiyagarajan, N. / Ferguson, R. / Saha, S. / Pham, T. / Subramanian, V. / Acharya, K.R. | ||||||
 Citation Citation |  Journal: Nat.Commun. / Year: 2012 Journal: Nat.Commun. / Year: 2012Title: Structural and Molecular Insights Into the Mechanism of Action of Human Angiogenin-Als Variants in Neurons. Authors: Thiyagarajan, N. / Ferguson, R. / Subramanian, V. / Acharya, K.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4ahl.cif.gz 4ahl.cif.gz | 38.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4ahl.ent.gz pdb4ahl.ent.gz | 26.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4ahl.json.gz 4ahl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  4ahl_validation.pdf.gz 4ahl_validation.pdf.gz | 417.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  4ahl_full_validation.pdf.gz 4ahl_full_validation.pdf.gz | 435.3 KB | Display | |
| Data in XML |  4ahl_validation.xml.gz 4ahl_validation.xml.gz | 8.2 KB | Display | |
| Data in CIF |  4ahl_validation.cif.gz 4ahl_validation.cif.gz | 10.8 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ah/4ahl https://data.pdbj.org/pub/pdb/validation_reports/ah/4ahl ftp://data.pdbj.org/pub/pdb/validation_reports/ah/4ahl ftp://data.pdbj.org/pub/pdb/validation_reports/ah/4ahl | HTTPS FTP |
-Related structure data
| Related structure data |  4ahdC  4aheC  4ahfC  4ahgC  4ahhC  4ahiC  4ahjC  4ahkC  4ahmC  4ahnC  4aohC  1angS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 14185.079 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PET22B(PLUS) / Production host: HOMO SAPIENS (human) / Plasmid: PET22B(PLUS) / Production host:  References: UniProt: P03950, Hydrolases; Acting on ester bonds; Endoribonucleases producing 3'-phosphomonoesters |
|---|---|
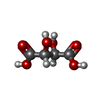
| #2: Chemical | ChemComp-TLA / |
| #3: Water | ChemComp-HOH / |
| Compound details | ENGINEERED |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.51 Å3/Da / Density % sol: 51.1 % / Description: NONE |
|---|---|
| Crystal grow | Details: 20 % PEG 4K, 0.4 M NA/K TARTRATE, 0.1 M NA CITRATE PH 5.5 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SRS SRS  / Beamline: PX10.1 / Wavelength: 0.9 / Beamline: PX10.1 / Wavelength: 0.9 |
| Detector | Type: ADSC CCD / Detector: CCD / Date: Oct 22, 2007 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9 Å / Relative weight: 1 |
| Reflection | Resolution: 2.05→50 Å / Num. obs: 9350 / % possible obs: 93 % / Observed criterion σ(I): 0 / Redundancy: 5.6 % / Biso Wilson estimate: 25.98 Å2 / Rmerge(I) obs: 0.1 / Net I/σ(I): 16.1 |
| Reflection shell | Resolution: 2.05→2.12 Å / Redundancy: 2.8 % / Rmerge(I) obs: 0.38 / Mean I/σ(I) obs: 2.5 / % possible all: 60.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1ANG Resolution: 2.053→25.44 Å / SU ML: 0.31 / σ(F): 0.04 / Phase error: 31.34 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 20.569 Å2 / ksol: 0.479 e/Å3 | ||||||||||||||||||||||||||||
| Displacement parameters |
| ||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.053→25.44 Å
| ||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller




























 PDBj
PDBj