+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1nz6 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Auxilin J-Domain | ||||||
 Components Components | Auxilin | ||||||
 Keywords Keywords |  PROTEIN BINDING / PROTEIN BINDING /  Alpha Helix / Anti-Parallel Helix Hairpin Alpha Helix / Anti-Parallel Helix Hairpin | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of clathrin coat assembly /  synaptic vesicle recycling / synaptic vesicle uncoating / clathrin heavy chain binding / clathrin coat disassembly / synaptic vesicle recycling / synaptic vesicle uncoating / clathrin heavy chain binding / clathrin coat disassembly /  Hydrolases; Acting on ester bonds; Phosphoric-monoester hydrolases / Hydrolases; Acting on ester bonds; Phosphoric-monoester hydrolases /  clathrin-dependent endocytosis / clathrin-dependent endocytosis /  clathrin-coated vesicle / clathrin-coated vesicle /  clathrin binding / clathrin binding /  intracellular transport ...regulation of clathrin coat assembly / intracellular transport ...regulation of clathrin coat assembly /  synaptic vesicle recycling / synaptic vesicle uncoating / clathrin heavy chain binding / clathrin coat disassembly / synaptic vesicle recycling / synaptic vesicle uncoating / clathrin heavy chain binding / clathrin coat disassembly /  Hydrolases; Acting on ester bonds; Phosphoric-monoester hydrolases / Hydrolases; Acting on ester bonds; Phosphoric-monoester hydrolases /  clathrin-dependent endocytosis / clathrin-dependent endocytosis /  clathrin-coated vesicle / clathrin-coated vesicle /  clathrin binding / clathrin binding /  intracellular transport / intracellular transport /  dephosphorylation / dephosphorylation /  heat shock protein binding / heat shock protein binding /  protein tyrosine phosphatase activity / protein tyrosine phosphatase activity /  SH3 domain binding / presynapse / vesicle / SH3 domain binding / presynapse / vesicle /  postsynaptic density / molecular adaptor activity / protein domain specific binding / intracellular membrane-bounded organelle / postsynaptic density / molecular adaptor activity / protein domain specific binding / intracellular membrane-bounded organelle /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Bos taurus (cattle) Bos taurus (cattle) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 2.5 Å MAD / Resolution: 2.5 Å | ||||||
 Authors Authors | Jiang, J. / Taylor, A.B. / Prasad, K. / Ishikawa-Brush, Y. / Hart, P.J. / Lafer, E.M. / Sousa, R. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2003 Journal: Biochemistry / Year: 2003Title: Structure-function analysis of the auxilin J-domain reveals an extended Hsc70 interaction interface. Authors: Jiang, J. / Taylor, A.B. / Prasad, K. / Ishikawa-Brush, Y. / Hart, P.J. / Lafer, E.M. / Sousa, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1nz6.cif.gz 1nz6.cif.gz | 47.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1nz6.ent.gz pdb1nz6.ent.gz | 38.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1nz6.json.gz 1nz6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nz/1nz6 https://data.pdbj.org/pub/pdb/validation_reports/nz/1nz6 ftp://data.pdbj.org/pub/pdb/validation_reports/nz/1nz6 ftp://data.pdbj.org/pub/pdb/validation_reports/nz/1nz6 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 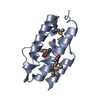
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein |  Mass: 12024.309 Da / Num. of mol.: 2 / Fragment: J-Domain Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Bos taurus (cattle) / Tissue: brain Bos taurus (cattle) / Tissue: brain / Plasmid: pGEX4T-2Aux813-910 / Production host: / Plasmid: pGEX4T-2Aux813-910 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q27974 Escherichia coli (E. coli) / References: UniProt: Q27974#2: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4 Å3/Da / Density % sol: 69.04 % | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | Temperature: 277 K / Method: microbatch under oil / pH: 8 Details: PEG 8000, Tris-Cl, TCEP, Glycerol, pH 8.0, microbatch under oil, temperature 277K | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / Method: batch method | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 113 K | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X8C / Wavelength: 0.9788, 0.9790, 0.9813 / Beamline: X8C / Wavelength: 0.9788, 0.9790, 0.9813 | ||||||||||||
| Detector | Type: RIGAKU RAXIS IV / Detector: IMAGE PLATE / Date: Oct 15, 2002 / Details: mirrors | ||||||||||||
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||
| Radiation wavelength |
| ||||||||||||
| Reflection | Resolution: 2.5→50 Å / Num. all: 13067 / Num. obs: 12866 / % possible obs: 98.5 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Rmerge(I) obs: 0.049 / Net I/σ(I): 38.9 | ||||||||||||
| Reflection shell | Highest resolution: 2.5 Å | ||||||||||||
| Reflection | *PLUS Lowest resolution: 50 Å / % possible obs: 100 % |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MAD / Resolution: 2.5→50 Å / σ(F): 0 / Stereochemistry target values: Engh & Huber MAD / Resolution: 2.5→50 Å / σ(F): 0 / Stereochemistry target values: Engh & Huber
| |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→50 Å
| |||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.5→2.59 Å /
| |||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 500 Å | |||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||
| Displacement parameters | *PLUS | |||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








