+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2cd0 | ||||||
|---|---|---|---|---|---|---|---|
| Title | STRUCTURE OF HUMAN LAMBDA-6 LIGHT CHAIN DIMER WIL | ||||||
 Components Components | PROTEIN (BENCE-JONES PROTEIN WIL, A VARIABLE DOMAIN FROM LAMBDA-6 TYPE IMMUNOGLOBULIN LIGHT CHAIN) | ||||||
 Keywords Keywords |  IMMUNE SYSTEM / IMMUNE SYSTEM /  IMMUNOGLOBULIN / IMMUNOGLOBULIN /  BENCE-JONES PROTEIN / BENCE-JONES PROTEIN /  LAMBDA-6 LAMBDA-6 | ||||||
| Function / homology |  Function and homology information Function and homology informationCD22 mediated BCR regulation / Fc epsilon receptor (FCERI) signaling /  immunoglobulin complex / Classical antibody-mediated complement activation / Initial triggering of complement / FCGR activation / Role of phospholipids in phagocytosis / Role of LAT2/NTAL/LAB on calcium mobilization / Scavenging of heme from plasma / FCERI mediated Ca+2 mobilization ...CD22 mediated BCR regulation / Fc epsilon receptor (FCERI) signaling / immunoglobulin complex / Classical antibody-mediated complement activation / Initial triggering of complement / FCGR activation / Role of phospholipids in phagocytosis / Role of LAT2/NTAL/LAB on calcium mobilization / Scavenging of heme from plasma / FCERI mediated Ca+2 mobilization ...CD22 mediated BCR regulation / Fc epsilon receptor (FCERI) signaling /  immunoglobulin complex / Classical antibody-mediated complement activation / Initial triggering of complement / FCGR activation / Role of phospholipids in phagocytosis / Role of LAT2/NTAL/LAB on calcium mobilization / Scavenging of heme from plasma / FCERI mediated Ca+2 mobilization / FCGR3A-mediated IL10 synthesis / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / immunoglobulin complex / Classical antibody-mediated complement activation / Initial triggering of complement / FCGR activation / Role of phospholipids in phagocytosis / Role of LAT2/NTAL/LAB on calcium mobilization / Scavenging of heme from plasma / FCERI mediated Ca+2 mobilization / FCGR3A-mediated IL10 synthesis / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers /  antigen binding / antigen binding /  Regulation of Complement cascade / Cell surface interactions at the vascular wall / FCERI mediated MAPK activation / FCGR3A-mediated phagocytosis / Regulation of actin dynamics for phagocytic cup formation / FCERI mediated NF-kB activation / Immunoregulatory interactions between a Lymphoid and a non-Lymphoid cell / Regulation of Complement cascade / Cell surface interactions at the vascular wall / FCERI mediated MAPK activation / FCGR3A-mediated phagocytosis / Regulation of actin dynamics for phagocytic cup formation / FCERI mediated NF-kB activation / Immunoregulatory interactions between a Lymphoid and a non-Lymphoid cell /  adaptive immune response / Potential therapeutics for SARS / adaptive immune response / Potential therapeutics for SARS /  immune response / immune response /  extracellular space / extracellular region / extracellular space / extracellular region /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å | ||||||
 Authors Authors | Pokkuluri, P.R. / Solomon, A. / Weiss, D.T. / Stevens, F.J. / Schiffer, M. | ||||||
 Citation Citation |  Journal: Amyloid / Year: 1999 Journal: Amyloid / Year: 1999Title: Tertiary structure of human lambda 6 light chains. Authors: Pokkuluri, P.R. / Solomon, A. / Weiss, D.T. / Stevens, F.J. / Schiffer, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2cd0.cif.gz 2cd0.cif.gz | 55.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2cd0.ent.gz pdb2cd0.ent.gz | 39.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2cd0.json.gz 2cd0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/cd/2cd0 https://data.pdbj.org/pub/pdb/validation_reports/cd/2cd0 ftp://data.pdbj.org/pub/pdb/validation_reports/cd/2cd0 ftp://data.pdbj.org/pub/pdb/validation_reports/cd/2cd0 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 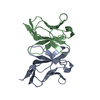 1cd0C  1dclS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Details | THE BIOLOGICALLY ACTIVE MOLECULE IS A HOMODIMER. THE ASYMMETRIC UNIT CONTAINS BOTH MONOMERS, CHAINS A AND B. BOTH MONOMERS REFINED INDEPENDENTLY. |
- Components
Components
| #1: Antibody | Mass: 12001.915 Da / Num. of mol.: 2 / Fragment: VARIABLE DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:   Escherichia coli (E. coli) / Strain (production host): DH5LAMBDA / References: Escherichia coli (E. coli) / Strain (production host): DH5LAMBDA / References:  GenBank: 587410, UniProt: P01721*PLUS GenBank: 587410, UniProt: P01721*PLUS#2: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2 Å3/Da / Density % sol: 38.5 % Description: CONSTANT DOMAINS WERE ALSO REMOVED FROM MCG MODEL PRIOR TO MOLECULAR REPLACEMENT. | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 5.6 Details: 1.8M AMMONIUM SULPHATE, 0.1 M SODIUM CITRATE PH 5.6, 0.01 M CADMIUM CHLORIDE | ||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion / PH range low: 8.5 / PH range high: 4.6 | ||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 19-ID / Wavelength: 1.033 / Beamline: 19-ID / Wavelength: 1.033 |
| Detector | Type: CUSTOM-MADE / Detector: CCD / Date: Sep 1, 1998 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.033 Å / Relative weight: 1 : 1.033 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→20 Å / Num. obs: 21478 / % possible obs: 97.6 % / Observed criterion σ(I): -3 / Redundancy: 21.8 % / Biso Wilson estimate: 16 Å2 / Rmerge(I) obs: 0.04 / Net I/σ(I): 48 |
| Reflection shell | Resolution: 1.7→1.76 Å / Redundancy: 9 % / Rmerge(I) obs: 0.232 / Mean I/σ(I) obs: 19 / % possible all: 96.4 |
| Reflection shell | *PLUS % possible obs: 96.4 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: MCG (1DCL): USED ONLY VL DIMER WITH CDR LOOPS DELETED. Resolution: 1.8→8 Å / Rfactor Rfree error: 0.009 / Data cutoff high rms absF: 1379583.8 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 3 / Details: BULK SOLVENT MODEL USED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 65 Å2 / ksol: 0.49 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 25.1 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.8→1.82 Å / Rfactor Rfree error: 0.054 / Total num. of bins used: 33
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: CNS / Version: 0.3 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller













 PDBj
PDBj




















