+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | PRD-0038 Spike glycoprotein NTD | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Sarbecoviruses /  Spike glycoprotein / Spike glycoprotein /  fusion protein / fusion protein /  neutralizing antibodies / neutralizing antibodies /  Structural Genomics / Seattle Structural Genomics Center for Infectious Disease / SSGCID / Structural Genomics / Seattle Structural Genomics Center for Infectious Disease / SSGCID /  inhibitor / inhibitor /  viral protein viral protein | |||||||||
| Biological species |  Sarbecovirus sp. Sarbecovirus sp. | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.9 Å cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Lee J / Park YJ / Veesler D | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Host Microbe / Year: 2023 Journal: Cell Host Microbe / Year: 2023Title: Broad receptor tropism and immunogenicity of a clade 3 sarbecovirus. Authors: Jimin Lee / Samantha K Zepeda / Young-Jun Park / Ashley L Taylor / Joel Quispe / Cameron Stewart / Elizabeth M Leaf / Catherine Treichel / Davide Corti / Neil P King / Tyler N Starr / David Veesler /   Abstract: Although Rhinolophus bats harbor diverse clade 3 sarbecoviruses, the structural determinants of receptor tropism along with the antigenicity of their spike (S) glycoproteins remain uncharacterized. ...Although Rhinolophus bats harbor diverse clade 3 sarbecoviruses, the structural determinants of receptor tropism along with the antigenicity of their spike (S) glycoproteins remain uncharacterized. Here, we show that the African Rhinolophus bat clade 3 sarbecovirus PRD-0038 S has a broad angiotensin-converting enzyme 2 (ACE2) usage and that receptor-binding domain (RBD) mutations further expand receptor promiscuity and enable human ACE2 utilization. We determine a cryo-EM structure of the PRD-0038 RBD bound to Rhinolophus alcyone ACE2, explaining receptor tropism and highlighting differences with SARS-CoV-1 and SARS-CoV-2. Characterization of PRD-0038 S using cryo-EM and monoclonal antibody reactivity reveals its distinct antigenicity relative to SARS-CoV-2 and identifies PRD-0038 cross-neutralizing antibodies for pandemic preparedness. PRD-0038 S vaccination elicits greater titers of antibodies cross-reacting with vaccine-mismatched clade 2 and clade 1a sarbecoviruses compared with SARS-CoV-2 S due to broader antigenic targeting, motivating the inclusion of clade 3 antigens in next-generation vaccines for enhanced resilience to viral evolution. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41843.map.gz emd_41843.map.gz | 483.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41843-v30.xml emd-41843-v30.xml emd-41843.xml emd-41843.xml | 16.1 KB 16.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41843.png emd_41843.png | 64.3 KB | ||
| Filedesc metadata |  emd-41843.cif.gz emd-41843.cif.gz | 5.4 KB | ||
| Others |  emd_41843_additional_1.map.gz emd_41843_additional_1.map.gz emd_41843_half_map_1.map.gz emd_41843_half_map_1.map.gz emd_41843_half_map_2.map.gz emd_41843_half_map_2.map.gz | 257.5 MB 475 MB 475 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41843 http://ftp.pdbj.org/pub/emdb/structures/EMD-41843 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41843 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41843 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41843.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41843.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_41843_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
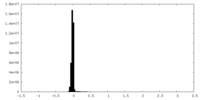
| Density Histograms |
-Half map: #1
| File | emd_41843_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
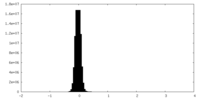
| Density Histograms |
-Half map: #2
| File | emd_41843_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
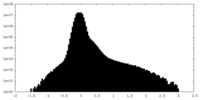
| Density Histograms |
- Sample components
Sample components
-Entire : PRD-0038 Spike glycoprotein
| Entire | Name: PRD-0038 Spike glycoprotein |
|---|---|
| Components |
|
-Supramolecule #1: PRD-0038 Spike glycoprotein
| Supramolecule | Name: PRD-0038 Spike glycoprotein / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Sarbecovirus sp. Sarbecovirus sp. |
-Macromolecule #1: PRD-0038 Spike glycoprotein NTD
| Macromolecule | Name: PRD-0038 Spike glycoprotein NTD / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Sarbecovirus sp. Sarbecovirus sp. |
| Sequence | String: MGILPSPGMP ALLSLVSLLS VLLMGCVAET GTQEGCGILS NKSKPALTQY SSSRRGFYYF DDTFRSSVRV LTTGYFLPFN SNLTGYSSRN AVTGRLIQFD NPNIPFKDGL YFAATERSNV IRGWIFGSTL DNTTQSAVLF NNGTHIVINV CNFYFCQDPM LTVANGSHYK ...String: MGILPSPGMP ALLSLVSLLS VLLMGCVAET GTQEGCGILS NKSKPALTQY SSSRRGFYYF DDTFRSSVRV LTTGYFLPFN SNLTGYSSRN AVTGRLIQFD NPNIPFKDGL YFAATERSNV IRGWIFGSTL DNTTQSAVLF NNGTHIVINV CNFYFCQDPM LTVANGSHYK SWVFLNATNC TYNRVHAFEI DPSLNTGAFI HLREHVFRNV DGFLYVYHNY ERANVYDNFP SGFSVLKPIL KLPFGLNITQ FKVIMTLFSP TTSSFNADAS VYFVGHLKPL TMLAEFDENG TITDAVDCSQ DPLSELKCTT KSLTVEKGIY QTSNFRVSPS TEVVRFPNIT NLCPFGQVFN ASKFPSVYAW ERLRISDCVA DYSVLYNSSS SFSTFKCYGV SPTKLNDLCF SSVYADYFVV KGDDVRQIAP AQTGVIADYN YKLPDDFTGC VLAWNTNSVD SKQGNNFYYR LFRHGKIKPY ERDISNVLYN SAGGTCSSTS QLGCYEPLKS YGFTPTVGVG YQPYRVVVLS FELLNAPATV CGPKKSTELV KNKCVNFNFN GLTGTGVLTS STKKFQPFQQ FGRDVSDFTD SVRDPKTLEI LDISPCSYGG VSVITPGTNT SKAVAVLYQD VNCTDVPTML HVEQVSTDWR VYALSADGNM FQTQAGCLVG ATYENSTYEC DIPIGAGICA KFGSNKLRLE SIVAYTMSIG EDQSIAYSNN TIAIPTNFSI SVTTEVLPVS MTKTSVDCNM YICGDSTECS NLLLQYGSFC TQLNRALSGI AVEQDRNTRD VFAQTKAIYK TPNIKDFGGF NFSQILPDPK KPSYRSPIED LLYNKVTLSD PGFMKQYGDC LGGINARDLI CAQKFNGLTV LPPLLTDDMI AAYTAALISG TATAGYTFGA GAALQIPFPM QMAYRFNGIG VTQNVLYENQ KQIANQFNNA ISKIQDSLTT TPAALGKLQD VINQNAVALN TLVKQLSSNF GAISSVLNDI LSRLDPPEAE VQIDRLITGR LQSLQTYVTQ QLIRAAEIRA SANLAATKMS ECVLGQSKRV DFCGKGYHLM SFPQAAPHGV VFLHVTYVPS QQQNFTTAPA ICHNGKAYFP REGVFVMNGT HWFITQRNFY SPQVITTDNT FESGSCDVVI GIVNNTVYDP LQPELESFKQ ELDKYFKNHT SPDVDLGDIS GINASVVDIK KEIAHLNEIA KNLNESLIDL QELGKYEQYV KSGRENLYFQ GGGGSGYIPE APRDGQAYVR KDGEWVLLST FLGHHHHHHH HGGSSGLNDI FEAQKIEWHE |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.0 µm Bright-field microscopy / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.0 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 2.9 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 310041 |
 Movie
Movie Controller
Controller









 Z
Z Y
Y X
X