+ Open data
Open data
- Basic information
Basic information
| Entry | 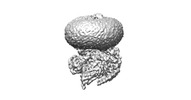 | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structures of peptide free and Gs-coupled GCGR | ||||||||||||||||||||||||
 Map data Map data | |||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
 Keywords Keywords |  G protein-coupled receptor / ligand recognition / G protein-coupled receptor / ligand recognition /  receptor activation / unimolecular agonist / receptor activation / unimolecular agonist /  MEMBRANE PROTEIN MEMBRANE PROTEIN | ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of glycogen metabolic process /  glucagon receptor activity / G-protein activation / Activation of the phototransduction cascade / : / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 ...regulation of glycogen metabolic process / glucagon receptor activity / G-protein activation / Activation of the phototransduction cascade / : / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 ...regulation of glycogen metabolic process /  glucagon receptor activity / G-protein activation / Activation of the phototransduction cascade / : / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Ca2+ pathway / Activation of G protein gated Potassium channels / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (z) signalling events / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Adrenaline,noradrenaline inhibits insulin secretion / ADP signalling through P2Y purinoceptor 12 / G alpha (q) signalling events / G alpha (i) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Glucagon-type ligand receptors / glucagon receptor activity / G-protein activation / Activation of the phototransduction cascade / : / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Ca2+ pathway / Activation of G protein gated Potassium channels / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (z) signalling events / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Adrenaline,noradrenaline inhibits insulin secretion / ADP signalling through P2Y purinoceptor 12 / G alpha (q) signalling events / G alpha (i) signalling events / Thrombin signalling through proteinase activated receptors (PARs) / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Glucagon-type ligand receptors /  alkylglycerophosphoethanolamine phosphodiesterase activity / Adrenaline,noradrenaline inhibits insulin secretion / G alpha (12/13) signalling events / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Thrombin signalling through proteinase activated receptors (PARs) / Ca2+ pathway / G alpha (z) signalling events / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / photoreceptor outer segment membrane / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / alkylglycerophosphoethanolamine phosphodiesterase activity / Adrenaline,noradrenaline inhibits insulin secretion / G alpha (12/13) signalling events / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Thrombin signalling through proteinase activated receptors (PARs) / Ca2+ pathway / G alpha (z) signalling events / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / photoreceptor outer segment membrane / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion /  spectrin binding / Vasopressin regulates renal water homeostasis via Aquaporins / response to starvation / cellular response to glucagon stimulus / spectrin binding / Vasopressin regulates renal water homeostasis via Aquaporins / response to starvation / cellular response to glucagon stimulus /  exocytosis / exocytosis /  peptide hormone binding / photoreceptor outer segment / cardiac muscle cell apoptotic process / photoreceptor inner segment / cellular response to starvation / hormone-mediated signaling pathway / guanyl-nucleotide exchange factor activity / response to nutrient / generation of precursor metabolites and energy / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Glucagon signaling in metabolic regulation / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Glucagon-type ligand receptors / peptide hormone binding / photoreceptor outer segment / cardiac muscle cell apoptotic process / photoreceptor inner segment / cellular response to starvation / hormone-mediated signaling pathway / guanyl-nucleotide exchange factor activity / response to nutrient / generation of precursor metabolites and energy / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / Glucagon signaling in metabolic regulation / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Glucagon-type ligand receptors /  regulation of blood pressure / cellular response to catecholamine stimulus / adenylate cyclase-activating dopamine receptor signaling pathway / cellular response to prostaglandin E stimulus / sensory perception of taste / G-protein beta-subunit binding / regulation of blood pressure / cellular response to catecholamine stimulus / adenylate cyclase-activating dopamine receptor signaling pathway / cellular response to prostaglandin E stimulus / sensory perception of taste / G-protein beta-subunit binding /  heterotrimeric G-protein complex / signaling receptor complex adaptor activity / heterotrimeric G-protein complex / signaling receptor complex adaptor activity /  glucose homeostasis / retina development in camera-type eye / glucose homeostasis / retina development in camera-type eye /  GTPase binding / GTPase binding /  cell body / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / cellular response to hypoxia / G alpha (s) signalling events / G alpha (q) signalling events / cell population proliferation / cell surface receptor signaling pathway / cell body / phospholipase C-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / cellular response to hypoxia / G alpha (s) signalling events / G alpha (q) signalling events / cell population proliferation / cell surface receptor signaling pathway /  endosome / G protein-coupled receptor signaling pathway / endosome / G protein-coupled receptor signaling pathway /  GTPase activity / GTPase activity /  dendrite / protein-containing complex binding / positive regulation of gene expression / dendrite / protein-containing complex binding / positive regulation of gene expression /  membrane / membrane /  plasma membrane / plasma membrane /  cytoplasm cytoplasmSimilarity search - Function | ||||||||||||||||||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /   Rattus norvegicus (Norway rat) / Rattus norvegicus (Norway rat) /   Bos taurus (cattle) / synthetic construct (others) / Bos taurus (cattle) / synthetic construct (others) /   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) | ||||||||||||||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.71 Å cryo EM / Resolution: 2.71 Å | ||||||||||||||||||||||||
 Authors Authors | Cong ZT / Zhao FH / Li Y / Luo G / Zhou QT / Yang DH / Wang MW | ||||||||||||||||||||||||
| Funding support |  China, 7 items China, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Cell Discov / Year: 2024 Journal: Cell Discov / Year: 2024Title: Molecular features of the ligand-free GLP-1R, GCGR and GIPR in complex with G proteins. Authors: Zhaotong Cong / Fenghui Zhao / Yang Li / Gan Luo / Yiting Mai / Xianyue Chen / Yanyan Chen / Shi Lin / Xiaoqing Cai / Qingtong Zhou / Dehua Yang / Ming-Wei Wang /   Abstract: Class B1 G protein-coupled receptors (GPCRs) are important regulators of many physiological functions such as glucose homeostasis, which is mainly mediated by three peptide hormones, i.e., glucagon- ...Class B1 G protein-coupled receptors (GPCRs) are important regulators of many physiological functions such as glucose homeostasis, which is mainly mediated by three peptide hormones, i.e., glucagon-like peptide-1 (GLP-1), glucagon (GCG), and glucose-dependent insulinotropic polypeptide (GIP). They trigger a cascade of signaling events leading to the formation of an active agonist-receptor-G protein complex. However, intracellular signal transducers can also activate the receptor independent of extracellular stimuli, suggesting an intrinsic role of G proteins in this process. Here, we report cryo-electron microscopy structures of the human GLP-1 receptor (GLP-1R), GCG receptor (GCGR), and GIP receptor (GIPR) in complex with G proteins without the presence of cognate ligands. These ligand-free complexes share a similar intracellular architecture to those bound by endogenous peptides, in which, the G protein alone directly opens the intracellular binding cavity and rewires the extracellular orthosteric pocket to stabilize the receptor in a state unseen before. While the peptide-binding site is partially occupied by the inward folded transmembrane helix 6 (TM6)-extracellular loop 3 (ECL3) juncture of GIPR or a segment of GCGR ECL2, the extracellular portion of GLP-1R adopts a conformation close to the active state. Our findings offer valuable insights into the distinct activation mechanisms of these three important receptors. It is possible that in the absence of a ligand, the intracellular half of transmembrane domain is mobilized with the help of G protein, which in turn rearranges the extracellular half to form a transitional conformation, facilitating the entry of the peptide N-terminus. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37505.map.gz emd_37505.map.gz | 46.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37505-v30.xml emd-37505-v30.xml emd-37505.xml emd-37505.xml | 21.2 KB 21.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_37505.png emd_37505.png | 31 KB | ||
| Filedesc metadata |  emd-37505.cif.gz emd-37505.cif.gz | 6.4 KB | ||
| Others |  emd_37505_half_map_1.map.gz emd_37505_half_map_1.map.gz emd_37505_half_map_2.map.gz emd_37505_half_map_2.map.gz | 84.5 MB 84.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37505 http://ftp.pdbj.org/pub/emdb/structures/EMD-37505 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37505 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37505 | HTTPS FTP |
-Related structure data
| Related structure data |  8wg8MC  8wa3C  8wg7C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_37505.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37505.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.071 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_37505_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_37505_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of the human glucagon like peptide 1 receptor i...
| Entire | Name: Cryo-EM structure of the human glucagon like peptide 1 receptor in complex with tirzepatide and G protein |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of the human glucagon like peptide 1 receptor i...
| Supramolecule | Name: Cryo-EM structure of the human glucagon like peptide 1 receptor in complex with tirzepatide and G protein type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short
| Macromolecule | Name: Guanine nucleotide-binding protein G(s) subunit alpha isoforms short type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 41.793418 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MGCTLSAEDK AAVERSKMIE KQLQKDKQVY RATHRLLLLG ADNSGKSTIV KQMRIYHVNG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLSAEDK AAVERSKMIE KQLQKDKQVY RATHRLLLLG ADNSGKSTIV KQMRIYHVNG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTSGIFETK FQVDKVNFHM FDVGAQRDER RKWIQCFNDV TAIIFVVASS SYNRLQEALN DF KSIWNNR WLRTISVILF LNKQDLLAEK VLAGKSKIED YFPEFARYTT PEDATPEPGE DPRVTRAKYF IRDEFLRIST ASG DGRHYC YPHFTCSVDT ENARRVFNDC RDIIQRMHLR QYELL |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Rattus norvegicus (Norway rat) Rattus norvegicus (Norway rat) |
| Molecular weight | Theoretical: 40.226992 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MGSLLQSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD ...String: MGSLLQSELD QLRQEAEQLK NQIRDARKAC ADATLSQITN NIDPVGRIQM RTRRTLRGHL AKIYAMHWGT DSRLLVSASQ DGKLIIWDS YTTNKVHAIP LRSSWVMTCA YAPSGNYVAC GGLDNICSIY NLKTREGNVR VSRELAGHTG YLSCCRFLDD N QIVTSSGD TTCALWDIET GQQTTTFTGH TGDVMSLSLA PDTRLFVSGA CDASAKLWDV REGMCRQTFT GHESDINAIC FF PNGNAFA TGSDDATCRL FDLRADQELM TYSHDNIICG ITSVSFSKSG RLLLAGYDDF NCNVWDALKA DRAGVLAGHD NRV SCLGVT DDGMAVATGS WDSFLKIWNG SSGGGGSGGG GSSGVSGWRL FKKIS UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Bos taurus (cattle) Bos taurus (cattle) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: Nanobody-35
| Macromolecule | Name: Nanobody-35 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 15.343019 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MAQVQLQESG GGLVQPGGSL RLSCAASGFT FSNYKMNWVR QAPGKGLEWV SDISQSGASI SYTGSVKGRF TISRDNAKNT LYLQMNSLK PEDTAVYYCA RCPAPFTRDC FDVTSTTYAY RGQGTQVTVS SHHHHHHEPE A |
-Macromolecule #5: Glucagon receptor
| Macromolecule | Name: Glucagon receptor / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 46.812441 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: AQVMDFLFEK WKLYGDQCHH NLSLLPPPTE LVCNRTFDKY SCWPDTPANT TANISCPWYL PWHHKVQHRF VFKRCGPDGQ WVRGPRGQP WRDASQCQMD GEEIEVQKEV AKMYSSFQVM YTVGYSLSLG ALLLALAILG GLSKLHCTRN AIHANLFASF V LKASSVLV ...String: AQVMDFLFEK WKLYGDQCHH NLSLLPPPTE LVCNRTFDKY SCWPDTPANT TANISCPWYL PWHHKVQHRF VFKRCGPDGQ WVRGPRGQP WRDASQCQMD GEEIEVQKEV AKMYSSFQVM YTVGYSLSLG ALLLALAILG GLSKLHCTRN AIHANLFASF V LKASSVLV IDGLLRTRYS QKIGDDLSVS TWLSDGAVAG CRVAAVFMQY GIVANYCWLL VEGLYLHNLL GLATLPERSF FS LYLGIGW GAPMLFVVPW AVVKCLFENV QCWTSNDNMG FWWILRFPVF LAILINFFIF VRIVQLLVAK LRARQMHHTD YKF RLAKST LTLIPLLGVH EVVFAFVTDE HAQGTLRSAK LFFDLFLSSF QGLLVAVLYC FLNKEVQSEL RRRWHRWRLG KVLW EERNT SN UniProtKB:  Glucagon receptor Glucagon receptor |
-Macromolecule #6: PRO-PRO-PRO-PRO-PHE-SER-ASN-LEU-VAL-MET-ASP-ASP-LEU-LYS-ASN-LYS-LYS
| Macromolecule | Name: PRO-PRO-PRO-PRO-PHE-SER-ASN-LEU-VAL-MET-ASP-ASP-LEU-LYS-ASN-LYS-LYS type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Molecular weight | Theoretical: 1.943289 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: PPPPFSNLVM DDLKNKK |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source: OTHER |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.2 µm Bright-field microscopy / Nominal defocus max: 2.2 µm / Nominal defocus min: 1.2 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 80.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Initial angle assignment | Type: OTHER |
| Final angle assignment | Type: OTHER |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.71 Å / Resolution method: OTHER / Number images used: 1232018 |
 Movie
Movie Controller
Controller



















 Z
Z Y
Y X
X