[English] 日本語
 Yorodumi
Yorodumi- EMDB-29700: Cryo-EM imaging scaffold subunits A and B used to display KRAS G1... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM imaging scaffold subunits A and B used to display KRAS G12C complex with GDP | |||||||||
 Map data Map data | scaffold subunits A and B used to display KRAS G12C complex with GDP Scaffolding Scaffolding | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CryoEM imaging scaffold /  Cancer / Cancer /  GTPase / GTPase /  STRUCTURAL PROTEIN STRUCTURAL PROTEIN | |||||||||
| Function / homology |  Cobalamin adenosyltransferase-like / Corrinoid adenosyltransferase, PduO-type / Cobalamin adenosyltransferase-like / Corrinoid adenosyltransferase, PduO-type /  Cobalamin adenosyltransferase / Cobalamin adenosyltransferase-like superfamily / Cobalamin adenosyltransferase / Cobalamin adenosyltransferase-like superfamily /  transferase activity / transferase activity /  ATP binding / Cobalamin adenosyltransferase-like domain-containing protein ATP binding / Cobalamin adenosyltransferase-like domain-containing protein Function and homology information Function and homology information | |||||||||
| Biological species | synthetic construct (others) /    Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.2 Å cryo EM / Resolution: 2.2 Å | |||||||||
 Authors Authors | Castells-Graells R / Sawaya MR / Yeates TO | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Cryo-EM structure determination of small therapeutic protein targets at 3 Å-resolution using a rigid imaging scaffold. Authors: Roger Castells-Graells / Kyle Meador / Mark A Arbing / Michael R Sawaya / Morgan Gee / Duilio Cascio / Emma Gleave / Judit É Debreczeni / Jason Breed / Karoline Leopold / Ankoor Patel / ...Authors: Roger Castells-Graells / Kyle Meador / Mark A Arbing / Michael R Sawaya / Morgan Gee / Duilio Cascio / Emma Gleave / Judit É Debreczeni / Jason Breed / Karoline Leopold / Ankoor Patel / Dushyant Jahagirdar / Bronwyn Lyons / Sriram Subramaniam / Chris Phillips / Todd O Yeates /    Abstract: Cryoelectron microscopy (Cryo-EM) has enabled structural determination of proteins larger than about 50 kDa, including many intractable by any other method, but it has largely failed for smaller ...Cryoelectron microscopy (Cryo-EM) has enabled structural determination of proteins larger than about 50 kDa, including many intractable by any other method, but it has largely failed for smaller proteins. Here, we obtain structures of small proteins by binding them to a rigid molecular scaffold based on a designed protein cage, revealing atomic details at resolutions reaching 2.9 Å. We apply this system to the key cancer signaling protein KRAS (19 kDa in size), obtaining four structures of oncogenic mutational variants by cryo-EM. Importantly, a structure for the key G12C mutant bound to an inhibitor drug (AMG510) reveals significant conformational differences compared to prior data in the crystalline state. The findings highlight the promise of cryo-EM scaffolds for advancing the design of drug molecules against small therapeutic protein targets in cancer and other human diseases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29700.map.gz emd_29700.map.gz | 86.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29700-v30.xml emd-29700-v30.xml emd-29700.xml emd-29700.xml | 16.7 KB 16.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_29700_fsc.xml emd_29700_fsc.xml | 9.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_29700.png emd_29700.png | 62.3 KB | ||
| Others |  emd_29700_half_map_1.map.gz emd_29700_half_map_1.map.gz emd_29700_half_map_2.map.gz emd_29700_half_map_2.map.gz | 84.7 MB 84.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29700 http://ftp.pdbj.org/pub/emdb/structures/EMD-29700 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29700 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29700 | HTTPS FTP |
-Related structure data
| Related structure data |  8g3kMC  8g42C  8g47C  8g4eC  8g4fC  8g4hC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_29700.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29700.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | scaffold subunits A and B used to display KRAS G12C complex with GDP | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half Map 1
| File | emd_29700_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 | ||||||||||||
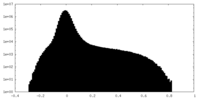
| Projections & Slices |
| ||||||||||||
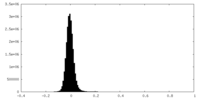
| Density Histograms |
-Half map: Half Map 2
| File | emd_29700_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 | ||||||||||||
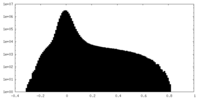
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM imaging scaffold displaying KRAS G12C
| Entire | Name: Cryo-EM imaging scaffold displaying KRAS G12C |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM imaging scaffold displaying KRAS G12C
| Supramolecule | Name: Cryo-EM imaging scaffold displaying KRAS G12C / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Macromolecule #1: Cob_adeno_trans domain-containing protein
| Macromolecule | Name: Cob_adeno_trans domain-containing protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:    Thermoplasma acidophilum (acidophilic) Thermoplasma acidophilum (acidophilic) |
| Molecular weight | Theoretical: 20.173395 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MRITTKVGDK GSTRLFGGEE VWKDDPIIEA NGTLDELTSF IGEAKHYVDE EMKGILEEIQ NDIYKIMGEI GSKGKIEGIS EERIKWLAG LIERYSEMVN KLSFVLPGGT LESAKLDVCR TIARRAERKV ATVLREFGIG TLAAIYLALL SRLLFLLARV I EIEKNKLK EVRSHHHHHH UniProtKB: Cobalamin adenosyltransferase-like domain-containing protein |
-Macromolecule #2: Cryo-EM imaging scaffold subunit B with DARPin - RCG-33
| Macromolecule | Name: Cryo-EM imaging scaffold subunit B with DARPin - RCG-33 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 35.452441 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MFTRRGDQGE TDLANRARVG KDSPVVEVQG TIDELNSFIG YALVLSRWDD IRNDLFRIQN DLFVLGEDVS TGGKGRTVTM DMIIYLIKR SVEMKAEIGK IELFVVPGGS VESASLHMAR AVSRRLERRI KAASELTEIN ANVLLYANML SNILFMHALI S NKRKEELD ...String: MFTRRGDQGE TDLANRARVG KDSPVVEVQG TIDELNSFIG YALVLSRWDD IRNDLFRIQN DLFVLGEDVS TGGKGRTVTM DMIIYLIKR SVEMKAEIGK IELFVVPGGS VESASLHMAR AVSRRLERRI KAASELTEIN ANVLLYANML SNILFMHALI S NKRKEELD KKLLEAARAG QDDEVAALLA KGADVNAHDT FGFTPLHLAA LYGHLEIVEV LLKRGADINA DDSYGRTPLH LA AMRGHLE IVELLLRWGA DVNAADEEGR TPLHLAAKRG HLEIVEVLLK NGADVNAQDK FGKTAFDISI DNGNEDLAEI LQK L |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.25 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 155000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.25 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 155000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8g3k: |
 Movie
Movie Controller
Controller







 Z
Z Y
Y X
X