[English] 日本語
 Yorodumi
Yorodumi- PDB-9sga: PENICILLIN-BINDING PROTEIN 1B (PBP-1B) IN COMPLEX WITH A MONOBACT... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9sga | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | PENICILLIN-BINDING PROTEIN 1B (PBP-1B) IN COMPLEX WITH A MONOBACTAM (21) | ||||||||||||||||||
 Components Components | Penicillin-binding protein 1B | ||||||||||||||||||
 Keywords Keywords | TRANSFERASE / CELL WALL / PEPTIDOGLYCAN SYNTHESIS ENZYME / DRUG-BINDING PROTEIN / MONOBACTAM | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationpeptidoglycan glycosyltransferase activity / serine-type D-Ala-D-Ala carboxypeptidase activity / penicillin binding / peptidoglycan biosynthetic process / cell wall organization / regulation of cell shape / outer membrane-bounded periplasmic space / proteolysis Similarity search - Function | ||||||||||||||||||
| Biological species |  | ||||||||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å MOLECULAR REPLACEMENT / Resolution: 1.5 Å | ||||||||||||||||||
 Authors Authors | Contreras-Martel, C. / Kavas, V. | ||||||||||||||||||
| Funding support |  Slovenia, Slovenia,  France, 5items France, 5items
| ||||||||||||||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2026 Journal: J.Med.Chem. / Year: 2026Title: Structure-Activity Relationship and Crystallographic Study of New Monobactams. Authors: Kavas, V. / Contreras-Martel, C. / Pajk, S. / Knez, D. / Martins, A. / Gould, T.A. / Roper, D.I. / Zdovc, I. / Dessen, A. / Hrast Rambaher, M. / Gobec, S. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9sga.cif.gz 9sga.cif.gz | 249.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9sga.ent.gz pdb9sga.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  9sga.json.gz 9sga.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sg/9sga https://data.pdbj.org/pub/pdb/validation_reports/sg/9sga ftp://data.pdbj.org/pub/pdb/validation_reports/sg/9sga ftp://data.pdbj.org/pub/pdb/validation_reports/sg/9sga | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  9sg5C  9sg6C  9sg7C  9sg8C  9sg9C  9sgbC  9sgcC  9sgdC  9sgeC C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 54085.879 Da / Num. of mol.: 1 / Mutation: N656G, R686Q, R687Q Source method: isolated from a genetically manipulated source Source: (gene. exp.)   | ||||||||
|---|---|---|---|---|---|---|---|---|---|
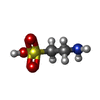
| #2: Chemical | ChemComp-A1JNU / [( Mass: 300.331 Da / Num. of mol.: 1 / Source method: obtained synthetically / Formula: C12H16N2O5S / Feature type: SUBJECT OF INVESTIGATION | ||||||||
| #3: Chemical | ChemComp-CL / #4: Chemical | ChemComp-TAU / | #5: Water | ChemComp-HOH / | Has ligand of interest | Y | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.31 Å3/Da / Density % sol: 62.85 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 7.2 Details: 50MM HEPES PH 7.2, 3M NACL, 0.6-0.9M AMMONIUM SULFATE |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: MASSIF-1 / Wavelength: 0.965459 Å / Beamline: MASSIF-1 / Wavelength: 0.965459 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Apr 19, 2024 |
| Radiation | Monochromator: C(110) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.965459 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→48.59 Å / Num. obs: 110122 / % possible obs: 96.6 % / Redundancy: 3.8 % / Biso Wilson estimate: 35.46 Å2 / CC1/2: 0.998 / Rsym value: 0.052 / Net I/σ(I): 10.26 |
| Reflection shell | Resolution: 1.5→1.59 Å / Redundancy: 3.7 % / Mean I/σ(I) obs: 0.75 / Num. unique obs: 17890 / CC1/2: 0.272 / Rsym value: 0.156 / % possible all: 98.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 1.5→48.582 Å / Cor.coef. Fo:Fc: 0.978 / Cor.coef. Fo:Fc free: 0.968 / SU B: 3.032 / SU ML: 0.051 / Cross valid method: FREE R-VALUE / ESU R: 0.054 / ESU R Free: 0.058 MOLECULAR REPLACEMENT / Resolution: 1.5→48.582 Å / Cor.coef. Fo:Fc: 0.978 / Cor.coef. Fo:Fc free: 0.968 / SU B: 3.032 / SU ML: 0.051 / Cross valid method: FREE R-VALUE / ESU R: 0.054 / ESU R Free: 0.058 Details: Hydrogens have been added in their riding positions
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK BULK SOLVENT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 35.035 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.5→48.582 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 20
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj