[English] 日本語
 Yorodumi
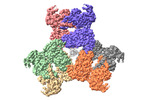
Yorodumi- PDB-8k04: CryoEM structure of a 2,3-hydroxycinnamic acid 1,2-dioxygenase Mh... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8k04 | ||||||
|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of a 2,3-hydroxycinnamic acid 1,2-dioxygenase MhpB in apo form | ||||||
 Components Components | 2,3-dihydroxyphenylpropionate/2,3-dihydroxicinnamic acid 1,2-dioxygenase | ||||||
 Keywords Keywords | BIOSYNTHETIC PROTEIN / dioxygenase | ||||||
| Function / homology | 3-carboxyethylcatechol 2,3-dioxygenase / 3-carboxyethylcatechol 2,3-dioxygenase activity / 2,3-dihydroxyphenylpropionate/2,3-dihydroxicinnamic acid 1,2-dioxygenase / Extradiol ring-cleavage dioxygenase, class III enzyme, subunit B / Catalytic LigB subunit of aromatic ring-opening dioxygenase / 3-phenylpropionate catabolic process / ferrous iron binding / 2,3-dihydroxyphenylpropionate/2,3-dihydroxicinnamic acid 1,2-dioxygenase Function and homology information Function and homology information | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.72 Å | ||||||
 Authors Authors | Jiang, W.X. / Cheng, X.Q. / Ma, L.X. / Xing, Q. | ||||||
| Funding support |  China, 1items China, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: CryoEM structure of the NADP-dependent malic enzyme in complex with oxaloacetate Authors: Jiang, W.X. / Cheng, X.Q. / Wu, M. / Ma, L.X. / Xing, Q. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8k04.cif.gz 8k04.cif.gz | 528.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8k04.ent.gz pdb8k04.ent.gz | 434 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8k04.json.gz 8k04.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8k04_validation.pdf.gz 8k04_validation.pdf.gz | 1.2 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8k04_full_validation.pdf.gz 8k04_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  8k04_validation.xml.gz 8k04_validation.xml.gz | 88.9 KB | Display | |
| Data in CIF |  8k04_validation.cif.gz 8k04_validation.cif.gz | 121.7 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/k0/8k04 https://data.pdbj.org/pub/pdb/validation_reports/k0/8k04 ftp://data.pdbj.org/pub/pdb/validation_reports/k0/8k04 ftp://data.pdbj.org/pub/pdb/validation_reports/k0/8k04 | HTTPS FTP |
-Related structure data
| Related structure data |  36757MC  8jzoC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: 1 / Ens-ID: ens_1 / Beg auth comp-ID: MET / Beg label comp-ID: MET / End auth comp-ID: ASN / End label comp-ID: ASN / Auth seq-ID: 1 - 314 / Label seq-ID: 1 - 314
NCS oper:
|
- Components
Components
| #1: Protein | Mass: 34230.961 Da / Num. of mol.: 10 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: mhpB, mhpB_1, A6592_08360, AAG43_003122, ACU57_18785, AM464_18695, AT845_003692, AW119_11075, BEA19_09385, BF481_002201, BG944_000993, BGM66_001457, BJI68_13680, BJJ90_20525, BKL28_002951, ...Gene: mhpB, mhpB_1, A6592_08360, AAG43_003122, ACU57_18785, AM464_18695, AT845_003692, AW119_11075, BEA19_09385, BF481_002201, BG944_000993, BGM66_001457, BJI68_13680, BJJ90_20525, BKL28_002951, BLM69_004076, BMC79_001945, BMT50_03125, BMT91_19310, BON92_03390, BTB68_001828, BUO55_000492, BvCmsKKP061_02674, BXT93_09690, BZL69_13825, C0P57_002705, C2R31_002140, C3F40_10810, C5N07_24670, C9E67_23975, CA593_01390, CDL36_14360, CDL37_09265, CG831_000406, CIG67_02690, CQ986_000779, CV83915_01226, CX938_004022, D0X26_07210, D4M65_16025, D9H94_11950, DN627_27135, DTL43_07625, DTL90_10525, DTM45_10750, DU321_19275, E4K51_23600, E5H86_16710, E6D34_13465, EA435_05985, EAN77_08135, EAX79_07015, EBP16_14625, ECs0403, EIZ93_16195, EL79_3496, ELT17_10045, ELT48_00575, ELX68_17755, ELX76_06030, ELX79_17290, EPS76_18260, ERS139208_01777, ExPECSC038_04860, F7F11_06665, F9413_17095, F9461_15720, F9S83_01345, FDM60_22665, FJQ53_15090, FKO60_15565, FOI11_011700, FOI11_08345, FPI65_01895, FV293_06300, FVB16_14265, FWK02_24950, FZU14_00995, G3565_02695, G3V95_15725, G4A38_05225, G4A47_03610, GF699_21980, GFY34_04780, GIB53_19760, GJ11_02180, GJO56_07880, GKF66_16190, GKF89_21475, GNW61_15375, GOP25_04445, GP944_04070, GP965_12675, GQM13_16935, GQM21_19370, GRC73_20955, GRW05_10415, GRW57_19650, GSM54_06355, GUI33_03835, H0O72_17150, HEP30_020365, HHH44_001337, HI055_001759, HIE29_000752, HJQ60_002399, HKA49_003062, HLV18_07550, HLX92_10820, HLZ50_17845, HMV95_12485, HV109_18305, HVY77_20140, HVZ29_04840, HX136_19710, IH772_19545, J4S20_003623, J5U05_003482, NCTC10418_05740, NCTC10429_03811, NCTC10764_05415, NCTC10974_04398, NCTC11126_05668, NCTC12950_04217, NCTC13127_05199, NCTC13148_06746, NCTC13216_02859, NCTC8179_01606, NCTC8333_04573, NCTC8450_01327, NCTC8622_02936, NCTC9044_03463, NCTC9045_04477, NCTC9073_03083, NCTC9077_04839, NCTC9111_04016, NCTC9962_02508, ND22_001410, SAMEA3472044_00503, SAMEA3472056_02987, SAMEA3751407_04552, SAMEA3752557_00177, SAMEA3753106_00568, WR15_20705 Production host:  References: UniProt: C3TMW2, 3-carboxyethylcatechol 2,3-dioxygenase |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Homohexamer of inositol phosphate phosphatase SopB / Type: COMPLEX / Entity ID: all / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  |
| Source (recombinant) | Organism: |
| Buffer solution | pH: 7.2 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: DIFFRACTION / Nominal defocus max: 1500 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 39 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3D reconstruction | Resolution: 2.72 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 364814 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 161.15 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj