[English] 日本語
 Yorodumi
Yorodumi- PDB-8e08: Crystal structure of HPSE P6 in complex with tetraose pentosan in... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 800000000 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of HPSE P6 in complex with tetraose pentosan inhibitor | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | HYDROLASE / Heparanase / Heparan Sulfate | |||||||||
| Function / homology |  Function and homology information Function and homology informationheparanase / heparanase activity / regulation of hair follicle development / heparin proteoglycan metabolic process / heparan sulfate proteoglycan catabolic process / beta-glucuronidase activity / HS-GAG degradation / positive regulation of hair follicle development / syndecan binding / proteoglycan metabolic process ...heparanase / heparanase activity / regulation of hair follicle development / heparin proteoglycan metabolic process / heparan sulfate proteoglycan catabolic process / beta-glucuronidase activity / HS-GAG degradation / positive regulation of hair follicle development / syndecan binding / proteoglycan metabolic process / vascular wound healing / protein transmembrane transport / establishment of endothelial barrier / angiogenesis involved in wound healing / positive regulation of osteoblast proliferation / positive regulation of vascular endothelial growth factor production / positive regulation of blood coagulation / lysosomal lumen / extracellular matrix / cell-matrix adhesion / specific granule lumen / lysosome / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / membrane raft / lysosomal membrane / response to antibiotic / intracellular membrane-bounded organelle / Neutrophil degranulation / extracellular space / extracellular region / nucleoplasm / nucleus Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.93 Å MOLECULAR REPLACEMENT / Resolution: 1.93 Å | |||||||||
 Authors Authors | Whitefield, C. / Jackson, C.J. | |||||||||
| Funding support |  Australia, 1items Australia, 1items
| |||||||||
 Citation Citation |  Journal: Biochemistry / Year: 2023 Journal: Biochemistry / Year: 2023Title: Complex Inhibitory Mechanism of Glycomimetics with Heparanase. Authors: Whitefield, C. / Vo, Y. / Schwartz, B.D. / Hepburn, C. / Ahmed, F.H. / Onagi, H. / Banwell, M.G. / Nelms, K. / Malins, L.R. / Jackson, C.J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8e08.cif.gz 8e08.cif.gz | 123.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8e08.ent.gz pdb8e08.ent.gz | 92.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8e08.json.gz 8e08.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/e0/8e08 https://data.pdbj.org/pub/pdb/validation_reports/e0/8e08 ftp://data.pdbj.org/pub/pdb/validation_reports/e0/8e08 ftp://data.pdbj.org/pub/pdb/validation_reports/e0/8e08 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 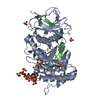 8e07C  7rg8S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 43375.863 Da / Num. of mol.: 1 Mutation: N178K, A195S, L197G, S212A, S219D, L230R, D234G, E244K, Q248H, R273G, S292A, R307L, I318T, S322Q, F327L, L354G, S426Q, K427D, K477Q, L483H, H486D, L498Q, M512K, E513P, S530A, A540P Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: HPSE, HEP, HPA, HPA1, HPR1, HPSE1, HSE1 / Production host: Homo sapiens (human) / Gene: HPSE, HEP, HPA, HPA1, HPR1, HPSE1, HSE1 / Production host:  |
|---|---|
| #2: Protein | Mass: 10267.545 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: HPSE, HEP, HPA, HPA1, HPR1, HPSE1, HSE1 / Production host: Homo sapiens (human) / Gene: HPSE, HEP, HPA, HPA1, HPR1, HPSE1, HSE1 / Production host:  |
-Sugars , 1 types, 3 molecules
| #3: Polysaccharide | Type: oligosaccharide / Mass: 1267.042 Da / Num. of mol.: 3 / Source method: obtained synthetically |
|---|
-Non-polymers , 3 types, 314 molecules 




| #4: Chemical | | #5: Chemical | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.59 Å3/Da / Density % sol: 52.59 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 5.5 Details: 0.2M Ammonium Sulfate, Sodium Acetate pH 5.5, 20% PEG 4000 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Australian Synchrotron Australian Synchrotron  / Beamline: MX2 / Wavelength: 0.9537 Å / Beamline: MX2 / Wavelength: 0.9537 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Dec 1, 2021 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9537 Å / Relative weight: 1 |
| Reflection | Resolution: 1.93→48.28 Å / Num. obs: 43216 / % possible obs: 100 % / Redundancy: 13 % / CC1/2: 0.997 / Rmerge(I) obs: 0.115 / Rpim(I) all: 0.033 / Rrim(I) all: 0.12 / Χ2: 0.75 / Net I/σ(I): 11.8 / Num. measured all: 561577 |
| Reflection shell | Resolution: 1.93→1.98 Å / % possible obs: 100 % / Redundancy: 13.7 % / Rmerge(I) obs: 0.925 / Num. measured all: 38828 / Num. unique obs: 2843 / CC1/2: 0.938 / Rpim(I) all: 0.257 / Rrim(I) all: 0.96 / Χ2: 0.39 / Net I/σ(I) obs: 3 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7RG8 Resolution: 1.93→48.28 Å / SU ML: 0.21 / Cross valid method: THROUGHOUT / σ(F): 1.35 / Phase error: 20.21 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.93→48.28 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj

