[English] 日本語
 Yorodumi
Yorodumi- PDB-8bts: Nitrogenase MoFe protein from A. vinelandii alpha double mutant C... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8bts | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Nitrogenase MoFe protein from A. vinelandii alpha double mutant C45A/L158C | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords | OXIDOREDUCTASE / N2-fixation / Nitrogenase / cooperativity / ammonia / hydrogen / metalloenzyme / FeMo-cofactor / P cluster / O2-sensitivity | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationnitrogen fixation / molybdenum-iron nitrogenase complex / nitrogenase / nitrogenase activity / iron-sulfur cluster binding / ATP binding / metal ion binding Similarity search - Function | |||||||||||||||
| Biological species |  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) | |||||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.03 Å MOLECULAR REPLACEMENT / Resolution: 3.03 Å | |||||||||||||||
 Authors Authors | Wagner, T. / Maslac, N. | |||||||||||||||
| Funding support |  Switzerland, Switzerland,  Germany, 4items Germany, 4items
| |||||||||||||||
 Citation Citation |  Journal: Jacs Au / Year: 2023 Journal: Jacs Au / Year: 2023Title: Nitrogen Fixation and Hydrogen Evolution by Sterically Encumbered Mo-Nitrogenase. Authors: Cadoux, C. / Ratcliff, D. / Maslac, N. / Gu, W. / Tsakoumagkos, I. / Hoogendoorn, S. / Wagner, T. / Milton, R.D. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8bts.cif.gz 8bts.cif.gz | 785.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8bts.ent.gz pdb8bts.ent.gz | 639.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8bts.json.gz 8bts.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bt/8bts https://data.pdbj.org/pub/pdb/validation_reports/bt/8bts ftp://data.pdbj.org/pub/pdb/validation_reports/bt/8bts ftp://data.pdbj.org/pub/pdb/validation_reports/bt/8bts | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
|---|---|
| Experimental dataset #1 | Data reference:  10.5281/zenodo.6865680 / Data set type: diffraction image data 10.5281/zenodo.6865680 / Data set type: diffraction image data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 2 types, 8 molecules ACHJBDIL
| #1: Protein | Mass: 56425.152 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Details: NifD containing two substitutions: C45A and L158C. / Source: (gene. exp.)  Azotobacter vinelandii DJ (bacteria) / Strain: RS1 / Tissue: / / Cell: / / Cell line: / / Gene: nifD / Organ: / / Variant: A. vinelandii nifD: C45A/L158C / Details (production host): / / Cell (production host): / / Cell line (production host): / / Organ (production host): / / Production host: Azotobacter vinelandii DJ (bacteria) / Strain: RS1 / Tissue: / / Cell: / / Cell line: / / Gene: nifD / Organ: / / Variant: A. vinelandii nifD: C45A/L158C / Details (production host): / / Cell (production host): / / Cell line (production host): / / Organ (production host): / / Production host:  Azotobacter vinelandii DJ (bacteria) / Strain (production host): RS1 / Tissue (production host): / / Variant (production host): A. vinelandii nifD: C45A/L158C / References: UniProt: C1DGZ7 Azotobacter vinelandii DJ (bacteria) / Strain (production host): RS1 / Tissue (production host): / / Variant (production host): A. vinelandii nifD: C45A/L158C / References: UniProt: C1DGZ7#2: Protein | Mass: 59535.879 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Azotobacter vinelandii DJ (bacteria) / Strain: RS1 / Tissue: / / Cell: / / Cell line: / / Gene: nifK / Organ: / / Variant: A. vinelandii nifD: C45A/L158C / Details (production host): / / Cell (production host): / / Cell line (production host): / / Organ (production host): / / Production host: Azotobacter vinelandii DJ (bacteria) / Strain: RS1 / Tissue: / / Cell: / / Cell line: / / Gene: nifK / Organ: / / Variant: A. vinelandii nifD: C45A/L158C / Details (production host): / / Cell (production host): / / Cell line (production host): / / Organ (production host): / / Production host:  Azotobacter vinelandii DJ (bacteria) / Strain (production host): RS1 / Tissue (production host): / / Variant (production host): A. vinelandii nifD: C45A/L158C / References: UniProt: P07329, nitrogenase Azotobacter vinelandii DJ (bacteria) / Strain (production host): RS1 / Tissue (production host): / / Variant (production host): A. vinelandii nifD: C45A/L158C / References: UniProt: P07329, nitrogenase |
|---|
-Non-polymers , 6 types, 38 molecules 
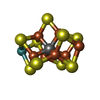




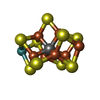




| #3: Chemical | ChemComp-HCA / #4: Chemical | ChemComp-ICS / #5: Chemical | ChemComp-GOL / #6: Chemical | ChemComp-SO4 / #7: Chemical | ChemComp-1CL / #8: Chemical | ChemComp-FE / |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.4 Å3/Da / Density % sol: 48.75 % / Description: Thin brown plate |
|---|---|
| Crystal grow | Temperature: 293.15 K / Method: vapor diffusion, sitting drop / pH: 5.5 Details: The purified enzyme in 50 mM Tris/HCl pH 8.0, 300 mM NaCl, 2 mM dithionite was crystallized at 8 mg.ml-1. Crystallization was performed anaerobically in a tent containing 100 % N2 atmosphere. ...Details: The purified enzyme in 50 mM Tris/HCl pH 8.0, 300 mM NaCl, 2 mM dithionite was crystallized at 8 mg.ml-1. Crystallization was performed anaerobically in a tent containing 100 % N2 atmosphere. Crystals were obtained by initial screening at 20 degree Celsius on 96-Well MRC 2-drop crystallization plates in polystyrene (SWISSCI) containing 90 ul of crystallization solution in the reservoir. The protein sample (0.5 ul) was mixed with 0.5 ul reservoir solution. The reservoir solution contained 25 % w/v Polyethylene glycol 3,350, 100 mM BIS-TRIS at pH 5.5, and 200 mM Lithium sulfate. The plate was then incubated in a Coy tent (N2/H2, 97:3) at 20 degree Celsius. Crystals appeared after a few weeks and were soaked in the crystallization solution supplemented with 15 % v/v glycerol as a cryo-protectant before being frozen in liquid nitrogen. PH range: / Temp details: Crystallized inside a Coy tent, a fluctuation of 1 degree might have occurred |
-Data collection
| Diffraction | Mean temperature: 100 K / Ambient temp details: / / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 1.00003 Å / Beamline: X06DA / Wavelength: 1.00003 Å |
| Detector | Type: DECTRIS PILATUS 2M-F / Detector: PIXEL / Date: Nov 27, 2021 / Details: / |
| Radiation | Monochromator: / / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.00003 Å / Relative weight: 1 |
| Reflection | Resolution: 3.029→143.493 Å / Num. obs: 39936 / % possible obs: 86.6 % / Redundancy: 3.7 % / CC1/2: 0.957 / Rmerge(I) obs: 0.297 / Rpim(I) all: 0.175 / Rrim(I) all: 0.346 / Net I/σ(I): 3.4 |
| Reflection shell | Resolution: 3.029→3.486 Å / Redundancy: 3.3 % / Rmerge(I) obs: 0.514 / Mean I/σ(I) obs: 1.6 / Num. unique obs: 1998 / CC1/2: 0.806 / Rpim(I) all: 0.334 / Rrim(I) all: 0.615 / % possible all: 69.4 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 3.03→95.5 Å / SU ML: 0.34 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 16.55 / Stereochemistry target values: ML MOLECULAR REPLACEMENT / Resolution: 3.03→95.5 Å / SU ML: 0.34 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 16.55 / Stereochemistry target values: MLDetails: No Translation-Libration-Screw-rotation, no non-crystallography symmetry, and no hydrogens were applied for the last cycle in Phenix.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 35.11 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.03→95.5 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj






