+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7uy1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | HUMAN PRMT5:MEP50 COMPLEX WITH MTA and Fragment 5 Bound | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSFERASE / MTAP / methyl transferase / fragment-based lead discovery / FBDD | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of adenylate cyclase-inhibiting dopamine receptor signaling pathway / peptidyl-arginine N-methylation / type II protein arginine methyltransferase / protein-arginine omega-N symmetric methyltransferase activity / peptidyl-arginine methylation / Golgi ribbon formation / negative regulation of epithelial cell proliferation involved in prostate gland development / oocyte axis specification / secretory columnal luminar epithelial cell differentiation involved in prostate glandular acinus development / histone H4R3 methyltransferase activity ...positive regulation of adenylate cyclase-inhibiting dopamine receptor signaling pathway / peptidyl-arginine N-methylation / type II protein arginine methyltransferase / protein-arginine omega-N symmetric methyltransferase activity / peptidyl-arginine methylation / Golgi ribbon formation / negative regulation of epithelial cell proliferation involved in prostate gland development / oocyte axis specification / secretory columnal luminar epithelial cell differentiation involved in prostate glandular acinus development / histone H4R3 methyltransferase activity / epithelial cell proliferation involved in prostate gland development / protein-arginine N-methyltransferase activity / methylosome / positive regulation of mRNA splicing, via spliceosome / methyl-CpG binding / endothelial cell activation / histone H3 methyltransferase activity / regulation of mitotic nuclear division / histone methyltransferase activity / negative regulation of gene expression via chromosomal CpG island methylation / Cul4B-RING E3 ubiquitin ligase complex / E-box binding / histone methyltransferase complex / positive regulation of oligodendrocyte differentiation / negative regulation of cell differentiation / spliceosomal snRNP assembly / ribonucleoprotein complex binding / regulation of ERK1 and ERK2 cascade / ubiquitin-like ligase-substrate adaptor activity / liver regeneration / regulation of signal transduction by p53 class mediator / methyltransferase activity / circadian regulation of gene expression / DNA-templated transcription termination / Regulation of TP53 Activity through Methylation / RMTs methylate histone arginines / protein polyubiquitination / p53 binding / transcription corepressor activity / snRNP Assembly / ubiquitin-dependent protein catabolic process / transcription coactivator activity / chromatin remodeling / protein heterodimerization activity / positive regulation of cell population proliferation / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / chromatin / Golgi apparatus / nucleoplasm / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.66 Å MOLECULAR REPLACEMENT / Resolution: 2.66 Å | ||||||
 Authors Authors | Gunn, R.J. | ||||||
| Funding support | 1items
| ||||||
 Citation Citation |  Journal: Rsc Med Chem / Year: 2022 Journal: Rsc Med Chem / Year: 2022Title: Fragment optimization and elaboration strategies - the discovery of two lead series of PRMT5/MTA inhibitors from five fragment hits. Authors: Smith, C.R. / Kulyk, S. / Ahmad, M.U.D. / Arkhipova, V. / Christensen, J.G. / Gunn, R.J. / Ivetac, A. / Ketcham, J.M. / Kuehler, J. / Lawson, J.D. / Thomas, N.C. / Wang, X. / Marx, M.A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7uy1.cif.gz 7uy1.cif.gz | 203.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7uy1.ent.gz pdb7uy1.ent.gz | 155.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7uy1.json.gz 7uy1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/uy/7uy1 https://data.pdbj.org/pub/pdb/validation_reports/uy/7uy1 ftp://data.pdbj.org/pub/pdb/validation_reports/uy/7uy1 ftp://data.pdbj.org/pub/pdb/validation_reports/uy/7uy1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7uyfC  7zupC  7zuqC  7zuuC  7zuyC  7zv2C  7zvlC  7zvuC  8csgC  8ctbC C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein , 2 types, 2 molecules AB
| #1: Protein | Mass: 73763.625 Da / Num. of mol.: 1 / Fragment: FULL LENGTH Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PRMT5, HRMT1L5, IBP72, JBP1, SKB1 / Production host: Homo sapiens (human) / Gene: PRMT5, HRMT1L5, IBP72, JBP1, SKB1 / Production host:  References: UniProt: O14744, type II protein arginine methyltransferase |
|---|---|
| #2: Protein | Mass: 37862.406 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: WDR77, MEP50, WD45, HKMT1069, Nbla10071 / Production host: Homo sapiens (human) / Gene: WDR77, MEP50, WD45, HKMT1069, Nbla10071 / Production host:  |
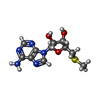
-Non-polymers , 7 types, 57 molecules 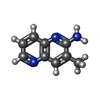











| #3: Chemical | ChemComp-PJ0 / | ||||||
|---|---|---|---|---|---|---|---|
| #4: Chemical | ChemComp-MTA / | ||||||
| #5: Chemical | ChemComp-SO4 / | ||||||
| #6: Chemical | | #7: Chemical | #8: Chemical | #9: Water | ChemComp-HOH / | |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.97 Å3/Da / Density % sol: 58.58 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion / Details: PEG/Buffer/Salt |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X10SA / Wavelength: 1 Å / Beamline: X10SA / Wavelength: 1 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Oct 17, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.66→45.96 Å / Num. obs: 34054 / % possible obs: 93.8 % / Redundancy: 5 % / Rmerge(I) obs: 0.05 / Net I/σ(I): 25.3 |
| Reflection shell | Resolution: 2.66→2.91 Å / Redundancy: 5.1 % / Rmerge(I) obs: 0.44 / Num. unique obs: 8304 / % possible all: 98.4 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: NONE Resolution: 2.66→45.96 Å / Cor.coef. Fo:Fc: 0.951 / Cor.coef. Fo:Fc free: 0.92 / SU B: 38.325 / SU ML: 0.345 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.801 / ESU R Free: 0.345 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 130.65 Å2 / Biso mean: 52 Å2 / Biso min: 33.88 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.66→45.96 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.66→2.729 Å / Rfactor Rfree error: 0
|
 Movie
Movie Controller
Controller



 PDBj
PDBj






