+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7sab | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Phencyclidine-bound GluN1a-GluN2B NMDA receptors | |||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||
 Keywords Keywords | TRANSPORT PROTEIN / Ligand-gated ion channel / ionotropic glutamate receptor / synaptic protein / voltage-gated ion channel | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationcellular response to corticosterone stimulus / cellular response to magnesium starvation / sensory organ development / cellular response to curcumin / regulation of cAMP/PKA signal transduction / pons maturation / EPHB-mediated forward signaling / Assembly and cell surface presentation of NMDA receptors / auditory behavior / positive regulation of Schwann cell migration ...cellular response to corticosterone stimulus / cellular response to magnesium starvation / sensory organ development / cellular response to curcumin / regulation of cAMP/PKA signal transduction / pons maturation / EPHB-mediated forward signaling / Assembly and cell surface presentation of NMDA receptors / auditory behavior / positive regulation of Schwann cell migration / regulation of cell communication / sensitization / olfactory learning / response to other organism / response to hydrogen sulfide / dendritic branch / fear response / conditioned taste aversion / response to methylmercury / protein localization to postsynaptic membrane / regulation of ARF protein signal transduction / apical dendrite / response to manganese ion / transmitter-gated monoatomic ion channel activity / suckling behavior / interleukin-1 receptor binding / response to carbohydrate / regulation of respiratory gaseous exchange / cellular response to lipid / propylene metabolic process / response to glycine / cellular response to dsRNA / RAF/MAP kinase cascade / negative regulation of dendritic spine maintenance / positive regulation of inhibitory postsynaptic potential / response to amine / response to growth hormone / heterocyclic compound binding / neurotransmitter receptor complex / Synaptic adhesion-like molecules / response to glycoside / regulation of monoatomic cation transmembrane transport / NMDA glutamate receptor activity / NMDA selective glutamate receptor complex / glutamate binding / voltage-gated monoatomic cation channel activity / ligand-gated sodium channel activity / neuromuscular process / regulation of axonogenesis / calcium ion transmembrane import into cytosol / positive regulation of glutamate secretion / regulation of dendrite morphogenesis / male mating behavior / regulation of synapse assembly / protein heterotetramerization / response to morphine / small molecule binding / glycine binding / receptor clustering / startle response / positive regulation of reactive oxygen species biosynthetic process / parallel fiber to Purkinje cell synapse / regulation of MAPK cascade / monoatomic ion channel complex / regulation of postsynaptic membrane potential / behavioral response to pain / monoatomic cation transmembrane transport / response to electrical stimulus / positive regulation of calcium ion transport into cytosol / extracellularly glutamate-gated ion channel activity / cellular response to glycine / associative learning / positive regulation of dendritic spine maintenance / action potential / response to magnesium ion / Unblocking of NMDA receptors, glutamate binding and activation / regulation of neuronal synaptic plasticity / monoatomic cation transport / glutamate receptor binding / detection of mechanical stimulus involved in sensory perception of pain / social behavior / ligand-gated monoatomic ion channel activity / multicellular organismal response to stress / response to mechanical stimulus / neuron development / phosphatase binding / long-term memory / prepulse inhibition / postsynaptic density, intracellular component / behavioral fear response / monoatomic cation channel activity / synaptic cleft / response to fungicide / calcium ion homeostasis / cellular response to manganese ion / glutamate-gated receptor activity / regulation of long-term synaptic depression / positive regulation of synaptic transmission, glutamatergic / glutamate-gated calcium ion channel activity / presynaptic active zone membrane Similarity search - Function | |||||||||||||||||||||
| Biological species |  | |||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 4.3 Å | |||||||||||||||||||||
 Authors Authors | Chou, T.-H. / Furukawa, H. | |||||||||||||||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||||||||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: Structural insights into binding of therapeutic channel blockers in NMDA receptors. Authors: Tsung-Han Chou / Max Epstein / Kevin Michalski / Eve Fine / Philip C Biggin / Hiro Furukawa /   Abstract: Excitatory signaling mediated by N-methyl-D-aspartate receptor (NMDAR) is critical for brain development and function, as well as for neurological diseases and disorders. Channel blockers of NMDARs ...Excitatory signaling mediated by N-methyl-D-aspartate receptor (NMDAR) is critical for brain development and function, as well as for neurological diseases and disorders. Channel blockers of NMDARs are of medical interest owing to their potential for treating depression, Alzheimer's disease, and epilepsy. However, precise mechanisms underlying binding and channel blockade have remained limited owing to challenges in obtaining high-resolution structures at the binding site within the transmembrane domains. Here, we monitor the binding of three clinically important channel blockers: phencyclidine, ketamine, and memantine in GluN1-2B NMDARs at local resolutions of 2.5-3.5 Å around the binding site using single-particle electron cryo-microscopy, molecular dynamics simulations, and electrophysiology. The channel blockers form different extents of interactions with the pore-lining residues, which control mostly off-speeds but not on-speeds. Our comparative analyses of the three unique NMDAR channel blockers provide a blueprint for developing therapeutic compounds with minimal side effects. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7sab.cif.gz 7sab.cif.gz | 547.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7sab.ent.gz pdb7sab.ent.gz | 434.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7sab.json.gz 7sab.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sa/7sab https://data.pdbj.org/pub/pdb/validation_reports/sa/7sab ftp://data.pdbj.org/pub/pdb/validation_reports/sa/7sab ftp://data.pdbj.org/pub/pdb/validation_reports/sa/7sab | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  24947MC  7saaC  7sacC  7sadC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
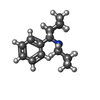
| #1: Protein | Mass: 95225.883 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Protein | Mass: 98888.945 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #3: Polysaccharide | Source method: isolated from a genetically manipulated source #4: Sugar | ChemComp-NAG / #5: Chemical | ChemComp-1PC / | Has ligand of interest | Y | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Hetero-tetrameric GluN1a-GluN2B NMDAR receptors / Type: COMPLEX / Entity ID: #1-#2 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: GOLD / Grid type: UltrAuFoil R1.2/1.3 |
| Vitrification | Cryogen name: ETHANE / Humidity: 85 % / Chamber temperature: 285 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 63 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.1_4122: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
| CTF correction | Type: NONE | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 4.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 163971 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






 PDBj
PDBj