[English] 日本語
 Yorodumi
Yorodumi- PDB-7ofs: Structure of SARS-CoV-2 Papain-like protease PLpro in complex wit... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7ofs | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of SARS-CoV-2 Papain-like protease PLpro in complex with 4-(2-hydroxyethyl)phenol | |||||||||
 Components Components | Papain-like protease nsp3 | |||||||||
 Keywords Keywords | HYDROLASE / Papain-like protease SARS-CoV-2 Hydrolase Zinc binding protein | |||||||||
| Function / homology |  Function and homology information Function and homology informationviral genome replication / methyltransferase activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / endonuclease activity / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / Translation of Replicase and Assembly of the Replication Transcription Complex / methylation / Replication of the SARS-CoV-2 genome ...viral genome replication / methyltransferase activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / endonuclease activity / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / Translation of Replicase and Assembly of the Replication Transcription Complex / methylation / Replication of the SARS-CoV-2 genome / double membrane vesicle viral factory outer membrane / SARS coronavirus main proteinase / host cell endosome / symbiont-mediated degradation of host mRNA / mRNA guanylyltransferase / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / mRNA guanylyltransferase activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / omega peptidase activity / SARS-CoV-2 modulates host translation machinery / symbiont-mediated perturbation of host ubiquitin-like protein modification / host cell Golgi apparatus / cysteine-type deubiquitinase activity / ubiquitinyl hydrolase 1 / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / single-stranded RNA binding / viral protein processing / host cell perinuclear region of cytoplasm / host cell endoplasmic reticulum membrane / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / symbiont-mediated suppression of host gene expression / viral translational frameshifting / symbiont-mediated activation of host autophagy / cysteine-type endopeptidase activity / lipid binding / host cell nucleus / SARS-CoV-2 activates/modulates innate and adaptive immune responses / proteolysis / zinc ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.9 Å MOLECULAR REPLACEMENT / Resolution: 1.9 Å | |||||||||
 Authors Authors | Srinivasan, V. / Werner, N. / Falke, S. / Guenther, S. / Reinke, P. / Ewert, W. / Sprenger, J. / Koua, F. / Brognaro, H. / Ullah, N. ...Srinivasan, V. / Werner, N. / Falke, S. / Guenther, S. / Reinke, P. / Ewert, W. / Sprenger, J. / Koua, F. / Brognaro, H. / Ullah, N. / Andaleeb, H. / Perbandt, M. / Alves Franca, B. / Schwinzer, M. / Wang, M. / Lieske, J. / Ginn, H. / Lane, T.J. / Yefanov, O. / Gelisio, L. / Hakanpaeae, J. / Saouane, S. / Tolstikova, A. / Groessler, M. / Fleckenstein, H. / Trost, F. / Wolf, M. / Lorenzen, K. / Schubert, R. / Han, H. / Schmidt, C. / Brings, L. / Galchenkova, M. / Gevorkov, Y. / Li, C. / Perk, A. / Awel, S. / Wahab, A. / Choudary, I. / Turk, D. / Hinrichs, W. / Chapman, H.N. / Meents, A. / Betzel, C. | |||||||||
| Funding support | European Union, 2items
| |||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2022 Journal: Commun Biol / Year: 2022Title: Antiviral activity of natural phenolic compounds in complex at an allosteric site of SARS-CoV-2 papain-like protease. Authors: Srinivasan, V. / Brognaro, H. / Prabhu, P.R. / de Souza, E.E. / Gunther, S. / Reinke, P.Y.A. / Lane, T.J. / Ginn, H. / Han, H. / Ewert, W. / Sprenger, J. / Koua, F.H.M. / Falke, S. / Werner, ...Authors: Srinivasan, V. / Brognaro, H. / Prabhu, P.R. / de Souza, E.E. / Gunther, S. / Reinke, P.Y.A. / Lane, T.J. / Ginn, H. / Han, H. / Ewert, W. / Sprenger, J. / Koua, F.H.M. / Falke, S. / Werner, N. / Andaleeb, H. / Ullah, N. / Franca, B.A. / Wang, M. / Barra, A.L.C. / Perbandt, M. / Schwinzer, M. / Schmidt, C. / Brings, L. / Lorenzen, K. / Schubert, R. / Machado, R.R.G. / Candido, E.D. / Oliveira, D.B.L. / Durigon, E.L. / Niebling, S. / Garcia, A.S. / Yefanov, O. / Lieske, J. / Gelisio, L. / Domaracky, M. / Middendorf, P. / Groessler, M. / Trost, F. / Galchenkova, M. / Mashhour, A.R. / Saouane, S. / Hakanpaa, J. / Wolf, M. / Alai, M.G. / Turk, D. / Pearson, A.R. / Chapman, H.N. / Hinrichs, W. / Wrenger, C. / Meents, A. / Betzel, C. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7ofs.cif.gz 7ofs.cif.gz | 155.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7ofs.ent.gz pdb7ofs.ent.gz | 118.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7ofs.json.gz 7ofs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/of/7ofs https://data.pdbj.org/pub/pdb/validation_reports/of/7ofs ftp://data.pdbj.org/pub/pdb/validation_reports/of/7ofs ftp://data.pdbj.org/pub/pdb/validation_reports/of/7ofs | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7nfvSC  7oftC  7ofuC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 35671.500 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Production host:  References: UniProt: P0DTC1, ubiquitinyl hydrolase 1, Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases |
|---|
-Non-polymers , 6 types, 212 molecules 

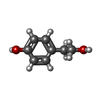








| #2: Chemical | ChemComp-ZN / | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #3: Chemical | | #4: Chemical | ChemComp-YRL / | #5: Chemical | ChemComp-PO4 / | #6: Chemical | ChemComp-GOL / | #7: Water | ChemComp-HOH / | |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.69 Å3/Da / Density % sol: 66.67 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop Details: Co-crystallization with the compounds was achieved mixing 0.2 uL of protein solution (22 mg/mL) in 50 mM TRIS buffer (pH 8.0) containing 1 mM TCEP and 150 mM NaCl with 0.1 uL of reservoir ...Details: Co-crystallization with the compounds was achieved mixing 0.2 uL of protein solution (22 mg/mL) in 50 mM TRIS buffer (pH 8.0) containing 1 mM TCEP and 150 mM NaCl with 0.1 uL of reservoir solution consisting of 1.0M NaH2PO4/1.0MKH2PO4, 100mM Tris_HCl pH=7.5. This growth solution was equilibrated by sitting drop vapor diffusion against 80 uL reservoir solution. Prior to crystallization 100 nL droplets of 10 mM compound solutions in DMSO were applied to the wells of SwissCI 96-well plates (2-well) and subsequently dried in vacuum. |
-Data collection
| Diffraction | Mean temperature: 277 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  PETRA III, DESY PETRA III, DESY  / Beamline: P11 / Wavelength: 1.0332 Å / Beamline: P11 / Wavelength: 1.0332 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Dec 16, 2020 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.0332 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→44.78 Å / Num. obs: 42250 / % possible obs: 99.92 % / Redundancy: 11 % / Biso Wilson estimate: 43.69 Å2 / CC1/2: 0.99 / Net I/σ(I): 15.2 |
| Reflection shell | Resolution: 1.9→1.968 Å / Redundancy: 10.5 % / Num. unique obs: 4154 / CC1/2: 0.505 / CC star: 0.819 / % possible all: 99.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7NFV Resolution: 1.9→44.78 Å / SU ML: 0.27 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 24.95 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 137.65 Å2 / Biso mean: 51.785 Å2 / Biso min: 30.2 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.9→44.78 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 15 / % reflection obs: 100 %
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 37.1827 Å / Origin y: 9.7339 Å / Origin z: 16.5612 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group | Selection details: (chain A and resseq 1:705) |
 Movie
Movie Controller
Controller














 PDBj
PDBj





