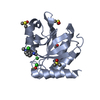
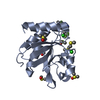
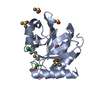
Entry Database : PDB / ID : 7h9dTitle Group deposition for crystallographic fragment screening of Chikungunya virus nsP3 macrodomain -- Crystal structure of Chikungunya virus nsP3 macrodomain in complex with Z728187792 (CHIKV_MacB-x1423) Non-structural protein 3 Keywords / / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / / / Resolution : 1.53 Å Authors Aschenbrenner, J.C. / Fairhead, M. / Godoy, A.S. / Balcomb, B.H. / Capkin, E. / Chandran, A.V. / Dolci, I. / Golding, M. / Koekemoer, L. / Lithgo, R.M. ...Aschenbrenner, J.C. / Fairhead, M. / Godoy, A.S. / Balcomb, B.H. / Capkin, E. / Chandran, A.V. / Dolci, I. / Golding, M. / Koekemoer, L. / Lithgo, R.M. / Marples, P.G. / Ni, X. / Oliva, G. / Thompson, W. / Tomlinson, C.W.E. / Wild, C. / Winokan, M. / Xavier, M.-A.E. / Fearon, D. / von Delft, F. Funding support Organization Grant number Country National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) U19AI171292
Journal : To Be Published Title : Group deposition for crystallographic fragment screening of Chikungunya virus nsP3 macrodomainAuthors: Aschenbrenner, J.C. / Fairhead, M. / Godoy, A.S. / Balcomb, B.H. / Capkin, E. / Chandran, A.V. / Dolci, I. / Golding, M. / Koekemoer, L. / Lithgo, R.M. / Marples, P.G. / Ni, X. / Oliva, G. / ... Authors : Aschenbrenner, J.C. / Fairhead, M. / Godoy, A.S. / Balcomb, B.H. / Capkin, E. / Chandran, A.V. / Dolci, I. / Golding, M. / Koekemoer, L. / Lithgo, R.M. / Marples, P.G. / Ni, X. / Oliva, G. / Thompson, W. / Tomlinson, C.W.E. / Wild, C. / Winokan, M. / Xavier, M.-A.E. / Fearon, D. / von Delft, F. History Deposition Apr 26, 2024 Deposition site / Processing site Revision 1.0 May 29, 2024 Provider / Type
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 Chikungunya virus
Chikungunya virus
 Chikungunya Virus
Chikungunya Virus X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.53 Å
MOLECULAR REPLACEMENT / Resolution: 1.53 Å  Authors
Authors United States, 1items
United States, 1items  Citation
Citation Journal: To Be Published
Journal: To Be Published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 7h9d.cif.gz
7h9d.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb7h9d.ent.gz
pdb7h9d.ent.gz PDB format
PDB format 7h9d.json.gz
7h9d.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/h9/7h9d
https://data.pdbj.org/pub/pdb/validation_reports/h9/7h9d ftp://data.pdbj.org/pub/pdb/validation_reports/h9/7h9d
ftp://data.pdbj.org/pub/pdb/validation_reports/h9/7h9d F&H Search
F&H Search Links
Links Assembly
Assembly




 Components
Components
 Chikungunya virus / Production host:
Chikungunya virus / Production host: 









 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Diamond
Diamond  / Beamline: I04-1 / Wavelength: 0.92124 Å
/ Beamline: I04-1 / Wavelength: 0.92124 Å Processing
Processing MOLECULAR REPLACEMENT / Resolution: 1.53→56.87 Å / Cor.coef. Fo:Fc: 0.967 / Cor.coef. Fo:Fc free: 0.956 / SU B: 4.451 / SU ML: 0.135 / Cross valid method: THROUGHOUT / ESU R: 0.115 / ESU R Free: 0.111 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
MOLECULAR REPLACEMENT / Resolution: 1.53→56.87 Å / Cor.coef. Fo:Fc: 0.967 / Cor.coef. Fo:Fc free: 0.956 / SU B: 4.451 / SU ML: 0.135 / Cross valid method: THROUGHOUT / ESU R: 0.115 / ESU R Free: 0.111 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS Movie
Movie Controller
Controller









 PDBj
PDBj




