+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6g29 | ||||||
|---|---|---|---|---|---|---|---|
| Title | X-ray structure of NSD3-PWWP1 in complex with compound 6 | ||||||
 Components Components | Histone-lysine N-methyltransferase NSD3 | ||||||
 Keywords Keywords | ONCOPROTEIN / Inhibitor / PWWP domain | ||||||
| Function / homology |  Function and homology information Function and homology information[histone H3]-lysine4 N-dimethyltransferase / [histone H3]-lysine27 N-dimethyltransferase / histone H3K4 dimethyltransferase activity / histone H3K27 dimethyltransferase activity / histone H3K27 trimethyltransferase activity / histone H3K36 methyltransferase activity / transcription regulator activator activity / histone H3 methyltransferase activity / PKMTs methylate histone lysines / methylation ...[histone H3]-lysine4 N-dimethyltransferase / [histone H3]-lysine27 N-dimethyltransferase / histone H3K4 dimethyltransferase activity / histone H3K27 dimethyltransferase activity / histone H3K27 trimethyltransferase activity / histone H3K36 methyltransferase activity / transcription regulator activator activity / histone H3 methyltransferase activity / PKMTs methylate histone lysines / methylation / regulation of DNA-templated transcription / positive regulation of DNA-templated transcription / chromatin / zinc ion binding / nucleoplasm / nucleus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 1.7 Å SYNCHROTRON / Resolution: 1.7 Å | ||||||
 Authors Authors | Boettcher, J. / Muellauer, B.J. / Weiss-Puxbaum, A. / Zoephel, A. | ||||||
 Citation Citation |  Journal: Nat.Chem.Biol. / Year: 2019 Journal: Nat.Chem.Biol. / Year: 2019Title: Fragment-based discovery of a chemical probe for the PWWP1 domain of NSD3. Authors: Bottcher, J. / Dilworth, D. / Reiser, U. / Neumuller, R.A. / Schleicher, M. / Petronczki, M. / Zeeb, M. / Mischerikow, N. / Allali-Hassani, A. / Szewczyk, M.M. / Li, F. / Kennedy, S. / ...Authors: Bottcher, J. / Dilworth, D. / Reiser, U. / Neumuller, R.A. / Schleicher, M. / Petronczki, M. / Zeeb, M. / Mischerikow, N. / Allali-Hassani, A. / Szewczyk, M.M. / Li, F. / Kennedy, S. / Vedadi, M. / Barsyte-Lovejoy, D. / Brown, P.J. / Huber, K.V.M. / Rogers, C.M. / Wells, C.I. / Fedorov, O. / Rumpel, K. / Zoephel, A. / Mayer, M. / Wunberg, T. / Bose, D. / Zahn, S. / Arnhof, H. / Berger, H. / Reiser, C. / Hormann, A. / Krammer, T. / Corcokovic, M. / Sharps, B. / Winkler, S. / Haring, D. / Cockcroft, X.L. / Fuchs, J.E. / Mullauer, B. / Weiss-Puxbaum, A. / Gerstberger, T. / Boehmelt, G. / Vakoc, C.R. / Arrowsmith, C.H. / Pearson, M. / McConnell, D.B. | ||||||
| History |
|
- Structure visualization
Structure visualization
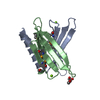
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6g29.cif.gz 6g29.cif.gz | 71.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6g29.ent.gz pdb6g29.ent.gz | 51.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6g29.json.gz 6g29.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/g2/6g29 https://data.pdbj.org/pub/pdb/validation_reports/g2/6g29 ftp://data.pdbj.org/pub/pdb/validation_reports/g2/6g29 ftp://data.pdbj.org/pub/pdb/validation_reports/g2/6g29 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6g24C  6g25C  6g27C  6g2bC  6g2cC  6g2eC  6g2fC  6g2oC  6g3pC  6g3tC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 16271.401 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: NSD3, WHSC1L1, DC28 / Production host: Homo sapiens (human) / Gene: NSD3, WHSC1L1, DC28 / Production host:  References: UniProt: Q9BZ95, histone-lysine N-methyltransferase |
|---|---|
| #2: Chemical | ChemComp-EHK / |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.28 Å3/Da / Density % sol: 46.05 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion Details: 100mM Morpheus Buffer 3, 30% P550MME_P20K, 10% Morpheus Ethylene glycols |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 1 Å / Beamline: X06DA / Wavelength: 1 Å |
| Detector | Type: DECTRIS PILATUS 2M-F / Detector: PIXEL / Date: Oct 17, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.697→38.17 Å / Num. obs: 15020 / % possible obs: 97.4 % / Redundancy: 6.4 % / Biso Wilson estimate: 29.13 Å2 / CC1/2: 1 / Rmerge(I) obs: 0.04 / Rpim(I) all: 0.017 / Rrim(I) all: 0.044 / Rsym value: 0.04 / Net I/σ(I): 24.5 |
| Reflection shell | Resolution: 1.697→1.703 Å / Redundancy: 6.4 % / Rmerge(I) obs: 0.85 / Mean I/σ(I) obs: 2.1 / Num. unique obs: 2193 / CC1/2: 0.814 / Rpim(I) all: 0.314 / Rrim(I) all: 0.812 / Rsym value: 0.85 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.7→38.17 Å / Cor.coef. Fo:Fc: 0.954 / Cor.coef. Fo:Fc free: 0.952 / SU R Cruickshank DPI: 0.113 / Cross valid method: THROUGHOUT / σ(F): 0 / SU R Blow DPI: 0.121 / SU Rfree Blow DPI: 0.105 / SU Rfree Cruickshank DPI: 0.101
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 125.28 Å2 / Biso mean: 38.36 Å2 / Biso min: 16.33 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.23 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.7→38.17 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.7→1.81 Å / Rfactor Rfree error: 0 / Total num. of bins used: 8
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: -8.343 Å / Origin y: 9.6866 Å / Origin z: -13.1781 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group | Selection details: { A|* } |
 Movie
Movie Controller
Controller












 PDBj
PDBj