+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6dq4 | ||||||
|---|---|---|---|---|---|---|---|
| Title | LINKED KDM5A JMJ DOMAIN BOUND TO THE INHIBITOR GSK-J1 | ||||||
 Components Components | Linked KDM5A Jmj Domain | ||||||
 Keywords Keywords | OXIDOREDUCTASE/INHIBITOR / DEMETHYLASE INHIBITION / OXIDOREDUCTASE-INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology information[histone H3]-trimethyl-L-lysine4 demethylase / histone H3K4me/H3K4me2/H3K4me3 demethylase activity / facultative heterochromatin formation / regulation of DNA-binding transcription factor activity / histone demethylase activity / enzyme inhibitor activity / Chromatin modifications during the maternal to zygotic transition (MZT) / HDMs demethylate histones / circadian regulation of gene expression / chromatin DNA binding ...[histone H3]-trimethyl-L-lysine4 demethylase / histone H3K4me/H3K4me2/H3K4me3 demethylase activity / facultative heterochromatin formation / regulation of DNA-binding transcription factor activity / histone demethylase activity / enzyme inhibitor activity / Chromatin modifications during the maternal to zygotic transition (MZT) / HDMs demethylate histones / circadian regulation of gene expression / chromatin DNA binding / protein-DNA complex / histone binding / transcription coactivator activity / transcription cis-regulatory region binding / chromatin remodeling / regulation of DNA-templated transcription / chromatin / positive regulation of DNA-templated transcription / nucleolus / negative regulation of transcription by RNA polymerase II / DNA binding / zinc ion binding / nucleoplasm / nucleus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.392 Å MOLECULAR REPLACEMENT / Resolution: 1.392 Å | ||||||
 Authors Authors | Horton, J.R. / Cheng, X. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: J. Med. Chem. / Year: 2018 Journal: J. Med. Chem. / Year: 2018Title: Structure-Based Engineering of Irreversible Inhibitors against Histone Lysine Demethylase KDM5A. Authors: Horton, J.R. / Woodcock, C.B. / Chen, Q. / Liu, X. / Zhang, X. / Shanks, J. / Rai, G. / Mott, B.T. / Jansen, D.J. / Kales, S.C. / Henderson, M.J. / Cyr, M. / Pohida, K. / Hu, X. / Shah, P. / ...Authors: Horton, J.R. / Woodcock, C.B. / Chen, Q. / Liu, X. / Zhang, X. / Shanks, J. / Rai, G. / Mott, B.T. / Jansen, D.J. / Kales, S.C. / Henderson, M.J. / Cyr, M. / Pohida, K. / Hu, X. / Shah, P. / Xu, X. / Jadhav, A. / Maloney, D.J. / Hall, M.D. / Simeonov, A. / Fu, H. / Vertino, P.M. / Cheng, X. #1:  Journal: Cell Chem Biol / Year: 2016 Journal: Cell Chem Biol / Year: 2016Title: Structural Basis for KDM5A Histone Lysine Demethylase Inhibition by Diverse Compounds. Authors: Horton, J.R. / Liu, X. / Gale, M. / Wu, L. / Shanks, J.R. / Zhang, X. / Webber, P.J. / Bell, J.S. / Kales, S.C. / Mott, B.T. / Rai, G. / Jansen, D.J. / Henderson, M.J. / Urban, D.J. / Hall, ...Authors: Horton, J.R. / Liu, X. / Gale, M. / Wu, L. / Shanks, J.R. / Zhang, X. / Webber, P.J. / Bell, J.S. / Kales, S.C. / Mott, B.T. / Rai, G. / Jansen, D.J. / Henderson, M.J. / Urban, D.J. / Hall, M.D. / Simeonov, A. / Maloney, D.J. / Johns, M.A. / Fu, H. / Jadhav, A. / Vertino, P.M. / Yan, Q. / Cheng, X. #2:  Journal: J. Biol. Chem. / Year: 2016 Journal: J. Biol. Chem. / Year: 2016Title: Characterization of a Linked Jumonji Domain of the KDM5/JARID1 Family of Histone H3 Lysine 4 Demethylases. Authors: Horton, J.R. / Engstrom, A. / Zoeller, E.L. / Liu, X. / Shanks, J.R. / Zhang, X. / Johns, M.A. / Vertino, P.M. / Fu, H. / Cheng, X. #3:  Journal: J. Med. Chem. / Year: 2018 Journal: J. Med. Chem. / Year: 2018Title: Insights into the Action of Inhibitor Enantiomers against Histone Lysine Demethylase 5A. Authors: Horton, J.R. / Liu, X. / Wu, L. / Zhang, K. / Shanks, J. / Zhang, X. / Rai, G. / Mott, B.T. / Jansen, D.J. / Kales, S.C. / Henderson, M.J. / Pohida, K. / Fang, Y. / Hu, X. / Jadhav, A. / ...Authors: Horton, J.R. / Liu, X. / Wu, L. / Zhang, K. / Shanks, J. / Zhang, X. / Rai, G. / Mott, B.T. / Jansen, D.J. / Kales, S.C. / Henderson, M.J. / Pohida, K. / Fang, Y. / Hu, X. / Jadhav, A. / Maloney, D.J. / Hall, M.D. / Simeonov, A. / Fu, H. / Vertino, P.M. / Yan, Q. / Cheng, X. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6dq4.cif.gz 6dq4.cif.gz | 152.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6dq4.ent.gz pdb6dq4.ent.gz | 116.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6dq4.json.gz 6dq4.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6dq4_validation.pdf.gz 6dq4_validation.pdf.gz | 814.2 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6dq4_full_validation.pdf.gz 6dq4_full_validation.pdf.gz | 817 KB | Display | |
| Data in XML |  6dq4_validation.xml.gz 6dq4_validation.xml.gz | 16.1 KB | Display | |
| Data in CIF |  6dq4_validation.cif.gz 6dq4_validation.cif.gz | 23.7 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dq/6dq4 https://data.pdbj.org/pub/pdb/validation_reports/dq/6dq4 ftp://data.pdbj.org/pub/pdb/validation_reports/dq/6dq4 ftp://data.pdbj.org/pub/pdb/validation_reports/dq/6dq4 | HTTPS FTP |
-Related structure data
| Related structure data |  6dq5C  6dq6C  6dq8C  6dq9C  6dqaC  6dqbC  5ivbS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 37944.836 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: KDM5A, JARID1A, RBBP2, RBP2 / Cell line (production host): GOLD C-PLUS / Production host: Homo sapiens (human) / Gene: KDM5A, JARID1A, RBBP2, RBP2 / Cell line (production host): GOLD C-PLUS / Production host:  References: UniProt: P29375, Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With 2-oxoglutarate as one donor, and incorporation of one atom of oxygen into each donor |
|---|
-Non-polymers , 6 types, 259 molecules 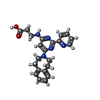










| #2: Chemical | ChemComp-K0I / | ||||||
|---|---|---|---|---|---|---|---|
| #3: Chemical | ChemComp-MN / | ||||||
| #4: Chemical | ChemComp-GOL / #5: Chemical | #6: Chemical | #7: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.26 Å3/Da / Density % sol: 45.59 % |
|---|---|
| Crystal grow | Temperature: 289 K / Method: vapor diffusion, sitting drop Details: 1.2-1.35 M (NH4)2SO4, 0.1 M Tris-HCl (pH 8.6-9.2) 0-20% glycerol 25 mM (Na/K) dibasic/monobasic phosphate PH range: 8.6-9.2 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-ID / Wavelength: 1 Å / Beamline: 22-ID / Wavelength: 1 Å |
| Detector | Type: RAYONIX MX300HE / Detector: CCD / Date: Mar 11, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.39→33.232 Å / Num. obs: 58786 / % possible obs: 86.9 % / Redundancy: 6.7 % / Rmerge(I) obs: 0.132 / Rpim(I) all: 0.051 / Net I/σ(I): 13.2 |
| Reflection shell | Resolution: 1.39→1.44 Å / Redundancy: 1.6 % / Rmerge(I) obs: 0.775 / Mean I/σ(I) obs: 1.3 / Num. unique obs: 2045 / CC1/2: 0.488 / Rpim(I) all: 0.605 / % possible all: 30.3 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5IVB Resolution: 1.392→33.232 Å / SU ML: 0.14 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 23.4
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.392→33.232 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj





