[English] 日本語
 Yorodumi
Yorodumi- PDB-6aox: F9 pilus adhesin FmlH lectin domain from E. coli UTI89 co-crystal... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6aox | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
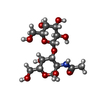
| Title | F9 pilus adhesin FmlH lectin domain from E. coli UTI89 co-crystallized with TF antigen | |||||||||
 Components Components | Fml fimbrial adhesin FmlD | |||||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / Fimbrial adhesin / lectin / bacterial adhesion | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | |||||||||
 Authors Authors | Kalas, V. / Hultgren, S.J. | |||||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2018 Journal: Proc.Natl.Acad.Sci.USA / Year: 2018Title: Structure-based discovery of glycomimetic FmlH ligands as inhibitors of bacterial adhesion during urinary tract infection. Authors: Kalas, V. / Hibbing, M.E. / Maddirala, A.R. / Chugani, R. / Pinkner, J.S. / Mydock-McGrane, L.K. / Conover, M.S. / Janetka, J.W. / Hultgren, S.J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6aox.cif.gz 6aox.cif.gz | 122.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6aox.ent.gz pdb6aox.ent.gz | 94.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6aox.json.gz 6aox.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ao/6aox https://data.pdbj.org/pub/pdb/validation_reports/ao/6aox ftp://data.pdbj.org/pub/pdb/validation_reports/ao/6aox ftp://data.pdbj.org/pub/pdb/validation_reports/ao/6aox | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6aowSC  6aoyC  6armC  6arnC  6aroC  6as8C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 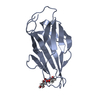
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 17920.088 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Polysaccharide | #3: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.94 Å3/Da / Density % sol: 36.56 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 5.6 Details: 0.2 M ammonium sulfate, 0.1M NaCl, 0.1 M MES pH 5.6, 32% PEG 3350 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 / Wavelength: 1.5418 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 / Wavelength: 1.5418 Å |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Apr 13, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.1→52.77 Å / Num. obs: 16562 / % possible obs: 99.6 % / Redundancy: 12 % / CC1/2: 0.994 / Rmerge(I) obs: 0.272 / Rpim(I) all: 0.08 / Rrim(I) all: 0.284 / Net I/σ(I): 8 |
| Reflection shell | Resolution: 2.1→2.21 Å / Redundancy: 12 % / Rmerge(I) obs: 1.887 / Mean I/σ(I) obs: 1.6 / Num. unique obs: 2379 / CC1/2: 0.393 / Rpim(I) all: 0.557 / Rrim(I) all: 1.97 / % possible all: 99.9 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6AOW Resolution: 2.1→52.77 Å / SU ML: 0.26 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 25.82
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→52.77 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj