[English] 日本語
 Yorodumi
Yorodumi- PDB-5wgl: Crystal structure of Danio rerio histone deacetylase 6 catalytic ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5wgl | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Danio rerio histone deacetylase 6 catalytic domain 2 in complex with ricolinostat (ACY-1215) | ||||||
 Components Components | Hdac6 protein | ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / histone deacetylase / hydrolase inhibitor / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationAggrephagy / negative regulation of cellular component organization / positive regulation of cellular component organization / deacetylase activity / tubulin deacetylase activity / mitochondrion localization / definitive hemopoiesis / regulation of microtubule-based process / protein lysine deacetylase activity / potassium ion binding ...Aggrephagy / negative regulation of cellular component organization / positive regulation of cellular component organization / deacetylase activity / tubulin deacetylase activity / mitochondrion localization / definitive hemopoiesis / regulation of microtubule-based process / protein lysine deacetylase activity / potassium ion binding / response to stress / hematopoietic progenitor cell differentiation / swimming behavior / transferase activity / chromatin organization / actin binding / angiogenesis / perikaryon / axon / centrosome / dendrite / zinc ion binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.7 Å MOLECULAR REPLACEMENT / Resolution: 1.7 Å | ||||||
 Authors Authors | Porter, N.J. / Christianson, D.W. | ||||||
 Citation Citation |  Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2017 Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2017Title: Unusual zinc-binding mode of HDAC6-selective hydroxamate inhibitors. Authors: Porter, N.J. / Mahendran, A. / Breslow, R. / Christianson, D.W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5wgl.cif.gz 5wgl.cif.gz | 174.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5wgl.ent.gz pdb5wgl.ent.gz | 134.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5wgl.json.gz 5wgl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/wg/5wgl https://data.pdbj.org/pub/pdb/validation_reports/wg/5wgl ftp://data.pdbj.org/pub/pdb/validation_reports/wg/5wgl ftp://data.pdbj.org/pub/pdb/validation_reports/wg/5wgl | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5wgiC  5wgkC  5wgmC  5eemS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 40285.484 Da / Num. of mol.: 2 / Fragment: catalytic domain 2 (UNP residues 288-646) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Non-polymers , 7 types, 575 molecules 

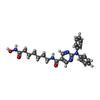










| #2: Chemical | | #3: Chemical | ChemComp-K / #4: Chemical | #5: Chemical | ChemComp-EDO / #6: Chemical | ChemComp-PEG / | #7: Chemical | ChemComp-FLC / | #8: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.83 Å3/Da / Density % sol: 56.59 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / Details: sodium citrate, pH 5.5, PEG8000 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 4.2.2 / Wavelength: 1.00003 Å / Beamline: 4.2.2 / Wavelength: 1.00003 Å |
| Detector | Type: RDI CMOS_8M / Detector: CMOS / Date: Sep 20, 2016 |
| Radiation | Monochromator: Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.00003 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→49.3 Å / Num. obs: 101104 / % possible obs: 99.9 % / Redundancy: 6.9 % / Net I/σ(I): 14.1 |
| Reflection shell | Highest resolution: 1.7 Å |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 5EEM Resolution: 1.7→49.298 Å / SU ML: 0.16 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 19.65
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.7→49.298 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj











