Entry Database : PDB / ID : 5p95Title rat catechol O-methyltransferase in complex with N-[5-(6-aminopurin-9-yl)pentyl]-5-(4-fluorophenyl)-2,3-dihydroxybenzamide at 1.30A Catechol O-methyltransferase Keywords / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Rattus norvegicus (Norway rat)Method / / / Resolution : 1.3 Å Authors Ehler, A. / Lerner, C. / Rudolph, M.G. Journal : To be published Title : Crystal Structure of a COMT complexAuthors : Lerner, C. / Rudolph, M.G. History Deposition Aug 30, 2016 Deposition site / Processing site Revision 1.0 Aug 30, 2017 Provider / Type Revision 2.0 Sep 27, 2017 Group Atomic model / Data collection ... Atomic model / Data collection / Derived calculations / Refinement description Category atom_site / atom_site_anisotrop ... atom_site / atom_site_anisotrop / refine / refine_hist / refine_ls_restr / refine_ls_shell / reflns / reflns_shell / software / struct_conn / struct_site / struct_site_gen Item _atom_site.label_alt_id / _atom_site_anisotrop.pdbx_label_alt_id ... _atom_site.label_alt_id / _atom_site_anisotrop.pdbx_label_alt_id / _refine.B_iso_max / _refine.B_iso_mean / _refine.B_iso_min / _refine.details / _refine.ls_R_factor_R_free / _refine.ls_R_factor_R_work / _refine.ls_percent_reflns_obs / _refine.pdbx_starting_model / _refine_hist.cycle_id / _refine_hist.number_atoms_solvent / _refine_hist.number_atoms_total / _refine_hist.pdbx_B_iso_mean_ligand / _refine_hist.pdbx_B_iso_mean_solvent / _refine_hist.pdbx_number_atoms_ligand / _refine_hist.pdbx_number_residues_total / _refine_ls_shell.d_res_high / _refine_ls_shell.d_res_low / _refine_ls_shell.number_reflns_all / _reflns.B_iso_Wilson_estimate / _reflns.pdbx_CC_half / _reflns.pdbx_Rrim_I_all / _reflns.pdbx_chi_squared / _reflns.pdbx_number_measured_all / _software.classification / _software.contact_author / _software.contact_author_email / _software.language / _software.location / _software.name / _software.type / _software.version Revision 2.1 Feb 21, 2018 Group / Category / Item Revision 2.2 Nov 17, 2021 Group / Derived calculations / Structure summaryCategory database_2 / pdbx_deposit_group ... database_2 / pdbx_deposit_group / struct_conn / struct_conn_type Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_deposit_group.group_description / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_ptnr2_label_alt_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn_type.id Revision 2.3 Apr 3, 2024 Group / Refinement descriptionCategory / chem_comp_bond / pdbx_initial_refinement_model
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3 Å
MOLECULAR REPLACEMENT / Resolution: 1.3 Å  Authors
Authors Citation
Citation Journal: To be published
Journal: To be published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5p95.cif.gz
5p95.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5p95.ent.gz
pdb5p95.ent.gz PDB format
PDB format 5p95.json.gz
5p95.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 5p95_validation.pdf.gz
5p95_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 5p95_full_validation.pdf.gz
5p95_full_validation.pdf.gz 5p95_validation.xml.gz
5p95_validation.xml.gz 5p95_validation.cif.gz
5p95_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/p9/5p95
https://data.pdbj.org/pub/pdb/validation_reports/p9/5p95 ftp://data.pdbj.org/pub/pdb/validation_reports/p9/5p95
ftp://data.pdbj.org/pub/pdb/validation_reports/p9/5p95 Links
Links Assembly
Assembly
 Components
Components




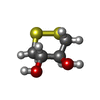

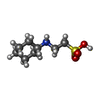







 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  SLS
SLS  / Beamline: X10SA / Wavelength: 1 Å
/ Beamline: X10SA / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller
























 PDBj
PDBj




