| Entry | Database: PDB / ID: 5owq
|
|---|
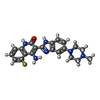
| Title | Human STK10 bound to dovitinib |
|---|
 Components Components | Serine/threonine-protein kinase 10 |
|---|
 Keywords Keywords | TRANSFERASE / Kinase / Structural Genomics Consortium / SGC |
|---|
| Function / homology |  Function and homology information Function and homology information
lymphocyte aggregation / regulation of lymphocyte migration / RHOB GTPase cycle / RHOC GTPase cycle / RHOA GTPase cycle / specific granule membrane / protein autophosphorylation / protein phosphorylation / non-specific serine/threonine protein kinase / intracellular signal transduction ...lymphocyte aggregation / regulation of lymphocyte migration / RHOB GTPase cycle / RHOC GTPase cycle / RHOA GTPase cycle / specific granule membrane / protein autophosphorylation / protein phosphorylation / non-specific serine/threonine protein kinase / intracellular signal transduction / nuclear body / protein serine kinase activity / protein serine/threonine kinase activity / Neutrophil degranulation / protein homodimerization activity / extracellular exosome / nucleoplasm / ATP binding / identical protein binding / plasma membrane / cytoplasm / cytosolSimilarity search - Function Serine/threonine-protein kinase 10, catalytic domain / Polo kinase kinase / : / Polo kinase kinase / Phosphorylase Kinase; domain 1 / Phosphorylase Kinase; domain 1 / Transferase(Phosphotransferase) domain 1 / Transferase(Phosphotransferase); domain 1 / Serine/threonine-protein kinase, active site / Serine/Threonine protein kinases active-site signature. ...Serine/threonine-protein kinase 10, catalytic domain / Polo kinase kinase / : / Polo kinase kinase / Phosphorylase Kinase; domain 1 / Phosphorylase Kinase; domain 1 / Transferase(Phosphotransferase) domain 1 / Transferase(Phosphotransferase); domain 1 / Serine/threonine-protein kinase, active site / Serine/Threonine protein kinases active-site signature. / Protein kinase domain / Serine/Threonine protein kinases, catalytic domain / Protein kinase, ATP binding site / Protein kinases ATP-binding region signature. / Protein kinase domain profile. / Protein kinase domain / Protein kinase-like domain superfamily / 2-Layer Sandwich / Orthogonal Bundle / Mainly Alpha / Alpha BetaSimilarity search - Domain/homology |
|---|
| Biological species |  Homo sapiens (human) Homo sapiens (human) |
|---|
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.7 Å MOLECULAR REPLACEMENT / Resolution: 2.7 Å |
|---|
 Authors Authors | Szklarz, M. / von Delft, F. / Bountra, C. / Knapp, S. / Edwards, A.M. / Arrowsmith, C. / Elkins, J.M. |
|---|
 Citation Citation |  Journal: To Be Published Journal: To Be Published
Title: Human STK10 bound to dovitinib
Authors: Elkins, J.M. |
|---|
| History | | Deposition | Sep 4, 2017 | Deposition site: PDBE / Processing site: PDBE |
|---|
| Revision 1.0 | Sep 13, 2017 | Provider: repository / Type: Initial release |
|---|
| Revision 1.1 | Jan 24, 2018 | Group: Source and taxonomy / Category: entity_src_gen / Item: _entity_src_gen.pdbx_host_org_scientific_name |
|---|
| Revision 1.2 | May 1, 2024 | Group: Data collection / Database references / Refinement description
Category: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_ncs_dom_lim
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession ..._database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id |
|---|
| Revision 1.3 | Oct 23, 2024 | Group: Structure summary / Category: pdbx_entry_details / pdbx_modification_feature |
|---|
|
|---|
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.7 Å
MOLECULAR REPLACEMENT / Resolution: 2.7 Å  Authors
Authors Citation
Citation Journal: To Be Published
Journal: To Be Published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5owq.cif.gz
5owq.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5owq.ent.gz
pdb5owq.ent.gz PDB format
PDB format 5owq.json.gz
5owq.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ow/5owq
https://data.pdbj.org/pub/pdb/validation_reports/ow/5owq ftp://data.pdbj.org/pub/pdb/validation_reports/ow/5owq
ftp://data.pdbj.org/pub/pdb/validation_reports/ow/5owq Links
Links Assembly
Assembly


 Components
Components Homo sapiens (human) / Gene: STK10, LOK / Plasmid: pNIC28-Bsa4 / Production host: Escherichia coli BL21(DE3)
Homo sapiens (human) / Gene: STK10, LOK / Plasmid: pNIC28-Bsa4 / Production host: Escherichia coli BL21(DE3)  X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Diamond
Diamond  / Beamline: I04-1 / Wavelength: 0.9173 Å
/ Beamline: I04-1 / Wavelength: 0.9173 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj