[English] 日本語
 Yorodumi
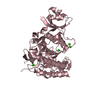
Yorodumi- PDB-5mrv: Crystal structure of human carboxypeptidase O in complex with NvCI -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mrv | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of human carboxypeptidase O in complex with NvCI | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE / Digestive Metallocarboxypeptidase / food digestion / carboxypeptidase inhibitor / Brush Border enzyme | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of endopeptidase activity / metalloendopeptidase inhibitor activity / Hydrolases; Acting on peptide bonds (peptidases); Metallocarboxypeptidases / side of membrane / metallocarboxypeptidase activity / apical plasma membrane / proteolysis / : / zinc ion binding / metal ion binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) Nerita versicolor (invertebrata) Nerita versicolor (invertebrata) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.854 Å MOLECULAR REPLACEMENT / Resolution: 1.854 Å | ||||||
 Authors Authors | Garcia-Pardo, J. / Garcia-Guerrero, M.C. / Fernandez-Alvarez, R. / Lyons, P. / Aviles, F.X. / Lorenzo, J. / Reverter, D. | ||||||
 Citation Citation |  Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2018 Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2018Title: Crystal structure and mechanism of human carboxypeptidase O: Insights into its specific activity for acidic residues. Authors: Garcia-Guerrero, M.C. / Garcia-Pardo, J. / Berenguer, E. / Fernandez-Alvarez, R. / Barfi, G.B. / Lyons, P.J. / Aviles, F.X. / Huber, R. / Lorenzo, J. / Reverter, D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mrv.cif.gz 5mrv.cif.gz | 163.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mrv.ent.gz pdb5mrv.ent.gz | 126.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mrv.json.gz 5mrv.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mr/5mrv https://data.pdbj.org/pub/pdb/validation_reports/mr/5mrv ftp://data.pdbj.org/pub/pdb/validation_reports/mr/5mrv ftp://data.pdbj.org/pub/pdb/validation_reports/mr/5mrv | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2pcuS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 40907.863 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CPO / Plasmid: pTriEx-7 vector / Cell line (production host): HEK293 F / Production host: Homo sapiens (human) / Gene: CPO / Plasmid: pTriEx-7 vector / Cell line (production host): HEK293 F / Production host:  Homo sapiens (human) Homo sapiens (human)References: UniProt: Q8IVL8, Hydrolases; Acting on peptide bonds (peptidases); Metallocarboxypeptidases #2: Protein | | Mass: 5956.682 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: Carboxypeptidase Inhibitor obtained from the marine snail Nerita versicolor Source: (gene. exp.)  Nerita versicolor (invertebrata) / Production host: Nerita versicolor (invertebrata) / Production host:  Komagataella phaffii GS115 (fungus) / References: UniProt: P86912 Komagataella phaffii GS115 (fungus) / References: UniProt: P86912#3: Chemical | #4: Sugar | ChemComp-NAG /   Type: D-saccharide, beta linking / Mass: 221.208 Da / Num. of mol.: 5 Type: D-saccharide, beta linking / Mass: 221.208 Da / Num. of mol.: 5Source method: isolated from a genetically manipulated source Formula: C8H15NO6 / Source: (gene. exp.)  Homo sapiens (human) / Gene: CPO / Plasmid: pTriEx-7 vector / Cell line (production host): HEK293 F / Production host: Homo sapiens (human) / Gene: CPO / Plasmid: pTriEx-7 vector / Cell line (production host): HEK293 F / Production host:  Homo sapiens (human) Homo sapiens (human)#5: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.74 Å3/Da / Density % sol: 55.08 % / Description: Monoclinic crystals C 1 2 1 |
|---|---|
| Crystal grow | Temperature: 291.15 K / Method: vapor diffusion, hanging drop / pH: 7 Details: Drops were prepared by mixing equal volumes of protein solution (CPO:NvCI molar ratio, 1:0.5) at 5 mg/ml (in 5 mM Tris-HCl pH 7.3, 100 mM NaCl, 1 mM B-mercaptoetanol) and reservoir solution ...Details: Drops were prepared by mixing equal volumes of protein solution (CPO:NvCI molar ratio, 1:0.5) at 5 mg/ml (in 5 mM Tris-HCl pH 7.3, 100 mM NaCl, 1 mM B-mercaptoetanol) and reservoir solution containning HEPES pH 7.0, 200 mM ammonium choride and 20% PEG6000. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALBA ALBA  / Beamline: XALOC / Wavelength: 0.9792 Å / Beamline: XALOC / Wavelength: 0.9792 Å |
| Detector | Type: DECTRIS PILATUS3 S 6M / Detector: PIXEL / Date: Oct 15, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9792 Å / Relative weight: 1 |
| Reflection | Resolution: 1.854→64.984 Å / Num. obs: 79948 / % possible obs: 98 % / Redundancy: 3.5 % / CC1/2: 0.998 / Rmerge(I) obs: 0.069 / Rsym value: 0.084 / Net I/σ(I): 13.9 |
| Reflection shell | Resolution: 1.854→1.86 Å / Redundancy: 3.4 % / Rmerge(I) obs: 0.8 / Mean I/σ(I) obs: 2 / CC1/2: 0.68 / % possible all: 96.7 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2PCU Resolution: 1.854→59.947 Å / SU ML: 0.23 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 23.19
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.854→59.947 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj



