[English] 日本語
 Yorodumi
Yorodumi- PDB-5mkf: cryoEM Structure of Polycystin-2 in complex with calcium and lipids -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mkf | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | cryoEM Structure of Polycystin-2 in complex with calcium and lipids | |||||||||
 Components Components | Polycystin-2 | |||||||||
 Keywords Keywords | TRANSPORT PROTEIN / Ca2+ signaling / cryoEM / membrane protein structure / Polycystin-2 / TRP channel | |||||||||
| Function / homology |  Function and homology information Function and homology informationdetection of nodal flow / metanephric smooth muscle tissue development / metanephric cortex development / metanephric cortical collecting duct development / metanephric distal tubule development / polycystin complex / mesonephric tubule development / mesonephric duct development / metanephric part of ureteric bud development / renal tubule morphogenesis ...detection of nodal flow / metanephric smooth muscle tissue development / metanephric cortex development / metanephric cortical collecting duct development / metanephric distal tubule development / polycystin complex / mesonephric tubule development / mesonephric duct development / metanephric part of ureteric bud development / renal tubule morphogenesis / determination of liver left/right asymmetry / metanephric ascending thin limb development / metanephric mesenchyme development / metanephric S-shaped body morphogenesis / basal cortex / renal artery morphogenesis / HLH domain binding / VxPx cargo-targeting to cilium / cilium organization / migrasome / muscle alpha-actinin binding / regulation of calcium ion import / calcium-induced calcium release activity / detection of mechanical stimulus / placenta blood vessel development / voltage-gated monoatomic ion channel activity / cellular response to hydrostatic pressure / cation channel complex / cellular response to fluid shear stress / non-motile cilium / outward rectifier potassium channel activity / motile cilium / cellular response to osmotic stress / determination of left/right symmetry / actinin binding / neural tube development / : / voltage-gated sodium channel activity / branching involved in ureteric bud morphogenesis / aorta development / voltage-gated monoatomic cation channel activity / ciliary membrane / negative regulation of G1/S transition of mitotic cell cycle / positive regulation of phospholipase C-activating G protein-coupled receptor signaling pathway / cytoplasmic side of endoplasmic reticulum membrane / protein heterotetramerization / heart looping / spinal cord development / centrosome duplication / embryonic placenta development / voltage-gated potassium channel activity / potassium channel activity / cell surface receptor signaling pathway via JAK-STAT / transcription regulator inhibitor activity / voltage-gated calcium channel activity / monoatomic cation channel activity / cytoskeletal protein binding / release of sequestered calcium ion into cytosol / potassium ion transmembrane transport / establishment of localization in cell / cellular response to calcium ion / basal plasma membrane / cytoplasmic vesicle membrane / cellular response to cAMP / sodium ion transmembrane transport / lumenal side of endoplasmic reticulum membrane / cellular response to reactive oxygen species / protein tetramerization / liver development / phosphoprotein binding / calcium ion transmembrane transport / Wnt signaling pathway / intracellular calcium ion homeostasis / positive regulation of nitric oxide biosynthetic process / cell-cell junction / calcium ion transport / mitotic spindle / regulation of cell population proliferation / lamellipodium / heart development / ATPase binding / protein homotetramerization / basolateral plasma membrane / transmembrane transporter binding / cell surface receptor signaling pathway / regulation of cell cycle / cilium / ciliary basal body / signaling receptor binding / negative regulation of cell population proliferation / calcium ion binding / positive regulation of gene expression / endoplasmic reticulum membrane / endoplasmic reticulum / Golgi apparatus / protein homodimerization activity / positive regulation of transcription by RNA polymerase II / extracellular exosome / membrane / identical protein binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 4.2 Å | |||||||||
 Authors Authors | Wilkes, M. / Madej, M.G. / Ziegler, C. | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2017 Journal: Nat Struct Mol Biol / Year: 2017Title: Molecular insights into lipid-assisted Ca regulation of the TRP channel Polycystin-2. Authors: Martin Wilkes / M Gregor Madej / Lydia Kreuter / Daniel Rhinow / Veronika Heinz / Silvia De Sanctis / Sabine Ruppel / Rebecca M Richter / Friederike Joos / Marina Grieben / Ashley C W Pike / ...Authors: Martin Wilkes / M Gregor Madej / Lydia Kreuter / Daniel Rhinow / Veronika Heinz / Silvia De Sanctis / Sabine Ruppel / Rebecca M Richter / Friederike Joos / Marina Grieben / Ashley C W Pike / Juha T Huiskonen / Elisabeth P Carpenter / Werner Kühlbrandt / Ralph Witzgall / Christine Ziegler /   Abstract: Polycystin-2 (PC2), a calcium-activated cation TRP channel, is involved in diverse Ca signaling pathways. Malfunctioning Ca regulation in PC2 causes autosomal-dominant polycystic kidney disease. Here ...Polycystin-2 (PC2), a calcium-activated cation TRP channel, is involved in diverse Ca signaling pathways. Malfunctioning Ca regulation in PC2 causes autosomal-dominant polycystic kidney disease. Here we report two cryo-EM structures of distinct channel states of full-length human PC2 in complex with lipids and cations. The structures reveal conformational differences in the selectivity filter and in the large exoplasmic domain (TOP domain), which displays differing N-glycosylation. The more open structure has one cation bound below the selectivity filter (single-ion mode, PC2), whereas multiple cations are bound along the translocation pathway in the second structure (multi-ion mode, PC2). Ca binding at the entrance of the selectivity filter suggests Ca blockage in PC2, and we observed density for the Ca-sensing C-terminal EF hand in the unblocked PC2 state. The states show altered interactions of lipids with the pore loop and TOP domain, thus reflecting the functional diversity of PC2 at different locations, owing to different membrane compositions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mkf.cif.gz 5mkf.cif.gz | 421.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mkf.ent.gz pdb5mkf.ent.gz | 321.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mkf.json.gz 5mkf.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mk/5mkf https://data.pdbj.org/pub/pdb/validation_reports/mk/5mkf ftp://data.pdbj.org/pub/pdb/validation_reports/mk/5mkf ftp://data.pdbj.org/pub/pdb/validation_reports/mk/5mkf | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3524MC  3523C  5mkeC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 109820.086 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PKD2, TRPP2 / Cell line (production host): HEK293 / Production host: Homo sapiens (human) / Gene: PKD2, TRPP2 / Cell line (production host): HEK293 / Production host:  Homo sapiens (human) / References: UniProt: Q13563 Homo sapiens (human) / References: UniProt: Q13563 |
|---|
-Sugars , 2 types, 16 molecules 
| #2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source #3: Sugar | ChemComp-NAG / |
|---|
-Non-polymers , 4 types, 29 molecules 

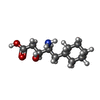




| #4: Chemical | ChemComp-PX6 / #5: Chemical | ChemComp-PLM / #6: Chemical | ChemComp-CHS / #7: Chemical | ChemComp-CA / |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Polycystin-2 / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Microscopy | Model: JEOL 3200FSC |
|---|---|
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Cs: 4.1 mm |
| Image recording | Electron dose: 1.8 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.10.1_2155: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Symmetry | Point symmetry: C4 (4 fold cyclic) | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 4.2 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 35318 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | Space: REAL Details: we used for comparative structure modeling TRPA1 (pdb entry code 3J9P) as template for S1 and S3-S5, TRPV1 (pdb entry code 3J5Q) for S5-S6, and the TRPV2 (pdb entry code 5AN8) fitted best ...Details: we used for comparative structure modeling TRPA1 (pdb entry code 3J9P) as template for S1 and S3-S5, TRPV1 (pdb entry code 3J5Q) for S5-S6, and the TRPV2 (pdb entry code 5AN8) fitted best for S2-S3 to obtain an initial model. The soluble domain was build based on pdbID: 5K47. But we had no search model for molecular replacement. Although we had a good idea what the architecture would be like, we build the model de novo with COOT. | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj










