[English] 日本語
 Yorodumi
Yorodumi- PDB-5l62: Yeast 20S proteasome with human beta5c (1-138) and human beta6 (9... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5l62 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Yeast 20S proteasome with human beta5c (1-138) and human beta6 (97-111; 118-133) in complex with epoxyketone inhibitor 16 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / HYDROLASE-HYDROLASE INHIBITOR complex / Proteasome / Mutant / Inhibitor / Binding Analysis | ||||||
| Function / homology |  Function and homology information Function and homology informationAntigen processing: Ub, ATP-independent proteasomal degradation / ER-Phagosome pathway / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome core complex assembly / Proteasome assembly / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / nuclear outer membrane-endoplasmic reticulum membrane network / Regulation of ornithine decarboxylase (ODC) / Regulation of PTEN stability and activity ...Antigen processing: Ub, ATP-independent proteasomal degradation / ER-Phagosome pathway / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome core complex assembly / Proteasome assembly / Cross-presentation of soluble exogenous antigens (endosomes) / TNFR2 non-canonical NF-kB pathway / nuclear outer membrane-endoplasmic reticulum membrane network / Regulation of ornithine decarboxylase (ODC) / Regulation of PTEN stability and activity / Proteasome assembly / CDK-mediated phosphorylation and removal of Cdc6 / proteasome core complex / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / Neddylation / Cross-presentation of soluble exogenous antigens (endosomes) / Somitogenesis / Ubiquitin-Mediated Degradation of Phosphorylated Cdc25A / Orc1 removal from chromatin / MAPK6/MAPK4 signaling / Antigen processing: Ubiquitination & Proteasome degradation / proteasome storage granule / proteasomal ubiquitin-independent protein catabolic process / Ub-specific processing proteases / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / endopeptidase activator activity / threonine-type endopeptidase activity / proteasome assembly / proteasome core complex, alpha-subunit complex / Neutrophil degranulation / : / proteasome complex / Regulation of activated PAK-2p34 by proteasome mediated degradation / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / Asymmetric localization of PCP proteins / Ubiquitin-dependent degradation of Cyclin D / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / AUF1 (hnRNP D0) binds and destabilizes mRNA / TNFR2 non-canonical NF-kB pathway / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of DVL / Degradation of AXIN / Degradation of CRY and PER proteins / Hh mutants are degraded by ERAD / Activation of NF-kappaB in B cells / G2/M Checkpoints / Degradation of GLI1 by the proteasome / Hedgehog ligand biogenesis / Regulation of RUNX3 expression and activity / Autodegradation of the E3 ubiquitin ligase COP1 / Defective CFTR causes cystic fibrosis / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / Negative regulation of NOTCH4 signaling / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / Hedgehog 'on' state / Vif-mediated degradation of APOBEC3G / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / Ubiquitin-Mediated Degradation of Phosphorylated Cdc25A / MAPK6/MAPK4 signaling / Degradation of CDH1 / Degradation of beta-catenin by the destruction complex / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / CDK-mediated phosphorylation and removal of Cdc6 / ABC-family protein mediated transport / CLEC7A (Dectin-1) signaling / SCF(Skp2)-mediated degradation of p27/p21 / FCERI mediated NF-kB activation / Regulation of expression of SLITs and ROBOs / Regulation of PTEN stability and activity / Interleukin-1 signaling / Orc1 removal from chromatin / Regulation of RAS by GAPs / Regulation of RUNX2 expression and activity / The role of GTSE1 in G2/M progression after G2 checkpoint / Separation of Sister Chromatids / UCH proteinases / KEAP1-NFE2L2 pathway / peroxisome / Downstream TCR signaling / peptidase activity / Antigen processing: Ubiquitination & Proteasome degradation / sperm principal piece / RUNX1 regulates transcription of genes involved in differentiation of HSCs / ER-Phagosome pathway / Neddylation / response to oxidative stress / sperm midpiece / secretory granule lumen / endopeptidase activity / ficolin-1-rich granule lumen / proteasome-mediated ubiquitin-dependent protein catabolic process Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.8 Å MOLECULAR REPLACEMENT / Resolution: 2.8 Å | ||||||
 Authors Authors | Groll, M. / Huber, E.M. | ||||||
| Funding support |  Germany, 1items Germany, 1items
| ||||||
 Citation Citation |  Journal: EMBO J. / Year: 2016 Journal: EMBO J. / Year: 2016Title: A humanized yeast proteasome identifies unique binding modes of inhibitors for the immunosubunit beta 5i. Authors: Huber, E.M. / Heinemeyer, W. / de Bruin, G. / Overkleeft, H.S. / Groll, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5l62.cif.gz 5l62.cif.gz | 2.4 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5l62.ent.gz pdb5l62.ent.gz | 2 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5l62.json.gz 5l62.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/l6/5l62 https://data.pdbj.org/pub/pdb/validation_reports/l6/5l62 ftp://data.pdbj.org/pub/pdb/validation_reports/l6/5l62 ftp://data.pdbj.org/pub/pdb/validation_reports/l6/5l62 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5l52C  5l54C  5l55C  5l5aC  5l5bC  5l5dC  5l5eC  5l5fC  5l5hC  5l5iC  5l5jC  5l5oC  5l5pC  5l5qC  5l5rC  5l5sC  5l5tC  5l5uC  5l5vC  5l5wC  5l5xC  5l5yC  5l5zC  5l60C  5l61C  5l63C  5l64C  5l65C  5l66C  5l67C  5l68C  5l69C  5l6aC  5l6bC  5l6cC  5lttC  5cz4S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS ensembles :
NCS oper:
|
- Components
Components
-Proteasome subunit alpha type- ... , 6 types, 12 molecules AOBPCQDRESGU
| #1: Protein | Mass: 27191.828 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P23639, proteasome endopeptidase complex #2: Protein | Mass: 28748.230 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P23638, proteasome endopeptidase complex #3: Protein | Mass: 28478.111 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P40303, proteasome endopeptidase complex #4: Protein | Mass: 28649.086 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P32379, proteasome endopeptidase complex #5: Protein | Mass: 25634.000 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P40302, proteasome endopeptidase complex #7: Protein | Mass: 28033.830 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P21243, proteasome endopeptidase complex |
|---|
-Protein , 1 types, 2 molecules FT
| #6: Protein | Mass: 31575.068 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P21242, proteasome endopeptidase complex |
|---|
-Proteasome subunit beta type- ... , 7 types, 14 molecules HVIWJXKYLZMaNb
| #8: Protein | Mass: 25114.459 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P25043, proteasome endopeptidase complex #9: Protein | Mass: 22627.842 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P25451, proteasome endopeptidase complex #10: Protein | Mass: 22545.676 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P22141, proteasome endopeptidase complex #11: Protein | Mass: 23177.207 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)  Gene: PSMB5, LMPX, MB1, X, PRE2, DOA3, PRG1, YPR103W, P8283.10 Production host:  References: UniProt: P28074, UniProt: P30656, proteasome endopeptidase complex #12: Protein | Mass: 24979.010 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)  Gene: PRE7, PRS3, PTS1, YBL041W, YBL0407, PSMB1, PSC5 / Production host:  References: UniProt: P23724, UniProt: P20618, proteasome endopeptidase complex #13: Protein | Mass: 27200.893 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P30657, proteasome endopeptidase complex #14: Protein | Mass: 21517.186 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P38624, proteasome endopeptidase complex |
|---|
-Non-polymers , 5 types, 311 molecules 


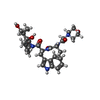





| #15: Chemical | ChemComp-MG / #16: Chemical | ChemComp-CL / #17: Chemical | ChemComp-MES / #18: Chemical | #19: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.67 Å3/Da / Density % sol: 66.52 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.8 / Details: 20 mM MgAC2, 13% MPD, 0.1 M MES / PH range: 6.8 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 1 Å / Beamline: X06SA / Wavelength: 1 Å |
| Detector | Type: PSI PILATUS 6M / Detector: PIXEL / Date: Jul 11, 2015 |
| Radiation | Monochromator: LN2 COOLED FIXED-EXIT. SI(111) MONOCHROMATOR / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.8→30 Å / Num. obs: 257546 / % possible obs: 97.7 % / Redundancy: 3.1 % / Rmerge(I) obs: 0.071 / Net I/σ(I): 13.9 |
| Reflection shell | Resolution: 2.8→2.9 Å / Rmerge(I) obs: 0.531 / Mean I/σ(I) obs: 2.8 / % possible all: 96.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5CZ4 Resolution: 2.8→15 Å / Cor.coef. Fo:Fc: 0.954 / Cor.coef. Fo:Fc free: 0.943 / SU B: 24.414 / SU ML: 0.205 / Cross valid method: THROUGHOUT / ESU R Free: 0.264 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 63.238 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.8→15 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj


















