[English] 日本語
 Yorodumi
Yorodumi- PDB-5ag1: DyP-type peroxidase of Auricularia auricula-judae (AauDyPI) with ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5ag1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | DyP-type peroxidase of Auricularia auricula-judae (AauDyPI) with meso- nitrated heme | |||||||||
 Components Components | DYE-DECOLORIZING PEROXIDASE | |||||||||
 Keywords Keywords | OXIDOREDUCTASE / DYP / FUNGAL / HEME / GLYCOPROTEIN / NITRATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationdye decolorizing peroxidase / lactoperoxidase activity / peroxidase / peroxidase activity / heme binding / extracellular region / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |  AURICULARIA AURICULA-JUDAE (jelly ear fungus) AURICULARIA AURICULA-JUDAE (jelly ear fungus) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.85 Å MOLECULAR REPLACEMENT / Resolution: 1.85 Å | |||||||||
 Authors Authors | Strittmatter, E. / Piontek, K. / Plattner, D.A. | |||||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Crystallographic Trapping of a Covalently Modified Heme in a Dye-Decolorizing Peroxidase Authors: Strittmatter, E. / Piontek, K. / Plattner, D.A. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5ag1.cif.gz 5ag1.cif.gz | 202.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5ag1.ent.gz pdb5ag1.ent.gz | 161 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5ag1.json.gz 5ag1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ag/5ag1 https://data.pdbj.org/pub/pdb/validation_reports/ag/5ag1 ftp://data.pdbj.org/pub/pdb/validation_reports/ag/5ag1 ftp://data.pdbj.org/pub/pdb/validation_reports/ag/5ag1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5ag0C  4au9S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 46805.855 Da / Num. of mol.: 2 / Fragment: DYP-TYPE PEROXIDASE DOMAIN, RESIDUES 64-509 / Source method: isolated from a natural source Details: GERMAN COLLECTION OF MICROORGANISMS (DSM), ACCESS NUMBER DSM 11326 Source: (natural)  AURICULARIA AURICULA-JUDAE (jelly ear fungus) AURICULARIA AURICULA-JUDAE (jelly ear fungus)Strain: SXM9-C021 / References: UniProt: I2DBY1, dye decolorizing peroxidase |
|---|
-Sugars , 2 types, 5 molecules 
| #2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source |
|---|---|
| #5: Sugar | ChemComp-NAG / |
-Non-polymers , 6 types, 908 molecules 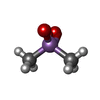


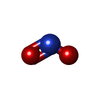






| #3: Chemical | | #4: Chemical | ChemComp-GOL / #6: Chemical | #7: Chemical | ChemComp-FMT / | #8: Chemical | #9: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y | ||
|---|---|---|---|
| Nonpolymer details | DELTA-MESO NITROHEME (N7H): IRON PROTOPORPH| Sequence details | PRECURSOR SEQUENCE IN GENBANK WITH PROLONGED N-TERMINUS. | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.2 Å3/Da / Density % sol: 44 % / Description: NONE |
|---|---|
| Crystal grow | pH: 6.5 Details: 0.17 M SODIUM ACETATE TRIHYDRATE, 0.085 M SODIUM CACODYLATE PH 6.5, 25% (W/V) PEG 8000, 15% (V/V) GLYCEROL |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-4 / Wavelength: 0.939 / Beamline: ID14-4 / Wavelength: 0.939 |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Jun 28, 2013 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.939 Å / Relative weight: 1 |
| Reflection | Resolution: 1.85→44.57 Å / Num. obs: 75411 / % possible obs: 98.2 % / Observed criterion σ(I): -3 / Redundancy: 3.2 % / Rmerge(I) obs: 0.1 / Net I/σ(I): 10.11 |
| Reflection shell | Resolution: 1.85→1.96 Å / Redundancy: 2.8 % / Rmerge(I) obs: 0.51 / Mean I/σ(I) obs: 2.1 / % possible all: 91.4 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 4AU9 Resolution: 1.85→44.57 Å / Cor.coef. Fo:Fc: 0.954 / Cor.coef. Fo:Fc free: 0.93 / SU B: 3.131 / SU ML: 0.093 / Cross valid method: THROUGHOUT / ESU R: 0.145 / ESU R Free: 0.135 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. U VALUES REFINED INDIVIDUALLY PROTEIN WAS TREATED WITH PROLI NONOATE AND PERACETIC ACID PRIOR TO CRYSTALLIZATION.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 16.212 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.85→44.57 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








