[English] 日本語
 Yorodumi
Yorodumi- PDB-3sbh: Crystal structure of human carbonic anhydrase isozyme II with 4-{... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3sbh | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of human carbonic anhydrase isozyme II with 4-{[(4,6-dimethyl-2-pyrimidinyl)sulfanyl]acetyl}benzenesulfonamide | ||||||
 Components Components | Carbonic anhydrase 2 | ||||||
 Keywords Keywords | lyase/lyase inhibitor / drug design / sulfonamide / metal-binding / lyase-lyase inhibitor complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of dipeptide transmembrane transport / : / regulation of monoatomic anion transport / secretion / cyanamide hydratase / cyanamide hydratase activity / arylesterase activity / regulation of chloride transport / Reversible hydration of carbon dioxide / morphogenesis of an epithelium ...positive regulation of dipeptide transmembrane transport / : / regulation of monoatomic anion transport / secretion / cyanamide hydratase / cyanamide hydratase activity / arylesterase activity / regulation of chloride transport / Reversible hydration of carbon dioxide / morphogenesis of an epithelium / angiotensin-activated signaling pathway / regulation of intracellular pH / Developmental Lineage of Pancreatic Ductal Cells / carbonic anhydrase / carbonate dehydratase activity / positive regulation of synaptic transmission, GABAergic / carbon dioxide transport / Erythrocytes take up oxygen and release carbon dioxide / Erythrocytes take up carbon dioxide and release oxygen / neuron cellular homeostasis / apical part of cell / myelin sheath / extracellular exosome / zinc ion binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  molecular replacement / Resolution: 1.65 Å molecular replacement / Resolution: 1.65 Å | ||||||
 Authors Authors | Grazulis, S. / Manakova, E. / Tamulaitiene, G. | ||||||
 Citation Citation |  Journal: Eur.J.Med.Chem. / Year: 2012 Journal: Eur.J.Med.Chem. / Year: 2012Title: Design of [(2-pyrimidinylthio)acetyl]benzenesulfonamides as inhibitors of human carbonic anhydrases. Authors: Capkauskaite, E. / Zubriene, A. / Baranauskiene, L. / Tamulaitiene, G. / Manakova, E. / Kairys, V. / Grazulis, S. / Tumkevicius, S. / Matulis, D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3sbh.cif.gz 3sbh.cif.gz | 78.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3sbh.ent.gz pdb3sbh.ent.gz | 56.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3sbh.json.gz 3sbh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sb/3sbh https://data.pdbj.org/pub/pdb/validation_reports/sb/3sbh ftp://data.pdbj.org/pub/pdb/validation_reports/sb/3sbh ftp://data.pdbj.org/pub/pdb/validation_reports/sb/3sbh | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3m2nC  3s8xC  3s9tC  3sapC  3saxC  3sbiC  3hljS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 29289.062 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CA2 / Plasmid: pET15b / Production host: Homo sapiens (human) / Gene: CA2 / Plasmid: pET15b / Production host:  |
|---|
-Non-polymers , 6 types, 309 molecules 
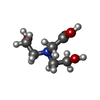

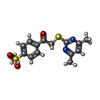







| #2: Chemical | ChemComp-ZN / | ||||||
|---|---|---|---|---|---|---|---|
| #3: Chemical | ChemComp-BCN / | ||||||
| #4: Chemical | | #5: Chemical | ChemComp-E65 / | #6: Chemical | ChemComp-MES / | #7: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.1 Å3/Da / Density % sol: 40.1 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 9 Details: 0.1M Na-bicine pH 9, 2M Na-malonate pH 7.55, VAPOR DIFFUSION, SITTING DROP, temperature 298K |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  EMBL/DESY, HAMBURG EMBL/DESY, HAMBURG  / Beamline: X11 / Wavelength: 0.8174 Å / Beamline: X11 / Wavelength: 0.8174 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: MAR555 FLAT PANEL / Detector: IMAGE PLATE / Date: Nov 14, 2010 / Details: Bent, vertically focussing | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: Si (111), horizontally focussing / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.8174 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.65→28.55 Å / Num. obs: 28165 / % possible obs: 97.7 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 7.6 % / Biso Wilson estimate: 16.2 Å2 / Rmerge(I) obs: 0.058 / Rsym value: 0.058 / Net I/σ(I): 30.7 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  molecular replacement molecular replacementStarting model: PDB entry 3HLJ Resolution: 1.65→25.23 Å / Cor.coef. Fo:Fc: 0.959 / Cor.coef. Fo:Fc free: 0.927 / WRfactor Rfree: 0.234 / WRfactor Rwork: 0.179 / Occupancy max: 1 / Occupancy min: 0 / FOM work R set: 0.849 / SU R Cruickshank DPI: 0.12 / SU Rfree: 0.124 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / ESU R Free: 0.124 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 56.28 Å2 / Biso mean: 15.461 Å2 / Biso min: 3.92 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.65→25.23 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.65→1.693 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller













 PDBj
PDBj






