[English] 日本語
 Yorodumi
Yorodumi- PDB-3qqs: Anthranilate phosphoribosyltransferase (TRPD) from Mycobacterium ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3qqs | ||||||
|---|---|---|---|---|---|---|---|
| Title | Anthranilate phosphoribosyltransferase (TRPD) from Mycobacterium tuberculosis (complex with inhibitor ACS172) | ||||||
 Components Components | Anthranilate phosphoribosyltransferase | ||||||
 Keywords Keywords | Transferase/Transferase Inhibitor / Anthranilate Phosphoribosyltransferase / Anthranilic Acids / Magnesium / Tryptophan / Magnesium binding / Phosphoribosyl pyrophosphate / Transferase-Transferase Inhibitor complex / Structural Genomics / TB Structural Genomics Consortium / TBSGC | ||||||
| Function / homology |  Function and homology information Function and homology informationanthranilate phosphoribosyltransferase / anthranilate phosphoribosyltransferase activity / L-tryptophan biosynthetic process / magnesium ion binding / extracellular region / plasma membrane / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.97 Å MOLECULAR REPLACEMENT / Resolution: 1.97 Å | ||||||
 Authors Authors | Castell, A. / Short, F.L. / Lott, J.S. / TB Structural Genomics Consortium (TBSGC) | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2013 Journal: Biochemistry / Year: 2013Title: The Substrate Capture Mechanism of Mycobacterium tuberculosis Anthranilate Phosphoribosyltransferase Provides a Mode for Inhibition. Authors: Castell, A. / Short, F.L. / Evans, G.L. / Cookson, T.V. / Bulloch, E.M. / Joseph, D.D. / Lee, C.E. / Parker, E.J. / Baker, E.N. / Lott, J.S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3qqs.cif.gz 3qqs.cif.gz | 269.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3qqs.ent.gz pdb3qqs.ent.gz | 217.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3qqs.json.gz 3qqs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qq/3qqs https://data.pdbj.org/pub/pdb/validation_reports/qq/3qqs ftp://data.pdbj.org/pub/pdb/validation_reports/qq/3qqs ftp://data.pdbj.org/pub/pdb/validation_reports/qq/3qqs | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3qr9C  3qs8C  3r6cC  3r88C  3twpC  3uu1C  1zvwS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | |
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Sugars , 2 types, 7 molecules ABCD

| #1: Protein | Mass: 38717.688 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: P66992, UniProt: P9WFX5*PLUS, anthranilate phosphoribosyltransferase #2: Sugar | |
|---|
-Non-polymers , 4 types, 614 molecules 
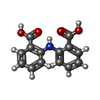
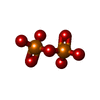




| #3: Chemical | ChemComp-MG / #4: Chemical | ChemComp-17C / #5: Chemical | ChemComp-DPO / | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.28 Å3/Da / Density % sol: 45.95 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 6.5 Details: 0.2 M cacodylate, 7% PEG6000, pH 6.5, VAPOR DIFFUSION, HANGING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 110 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: OTHER / Wavelength: 1.54179 Å ROTATING ANODE / Type: OTHER / Wavelength: 1.54179 Å |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Jun 4, 2009 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54179 Å / Relative weight: 1 |
| Reflection | Resolution: 1.97→94.9 Å / Num. obs: 97403 / % possible obs: 99.1 % / Observed criterion σ(F): 0 / Observed criterion σ(I): -3 / Redundancy: 3.7 % / Rmerge(I) obs: 0.091 / Rsym value: 0.106 |
| Reflection shell | Resolution: 1.97→2.08 Å / Redundancy: 3.6 % / Rmerge(I) obs: 0.72 / Mean I/σ(I) obs: 2 / Rsym value: 0.84 / % possible all: 97.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 1zvw Resolution: 1.97→88.75 Å / Cor.coef. Fo:Fc: 0.949 / Cor.coef. Fo:Fc free: 0.922 / Occupancy max: 1 / Occupancy min: 0.4 / SU B: 4.972 / SU ML: 0.14 / SU R Cruickshank DPI: 0.1949 / Cross valid method: THROUGHOUT / ESU R: 0.202 / ESU R Free: 0.177 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 26.272 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.97→88.75 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.97→2.021 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller
























 PDBj
PDBj




