+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2zrj | ||||||
|---|---|---|---|---|---|---|---|
| Title | MsRecA Q196A ATPgS form IV | ||||||
 Components Components | Protein recA | ||||||
 Keywords Keywords | HYDROLASE / recombination / RecA mutants / DNA-REPAIR / ATP-binding / DNA damage / DNA recombination / DNA repair / DNA-binding / Nucleotide-binding / SOS response | ||||||
| Function / homology |  Function and homology information Function and homology informationSOS response / ATP-dependent DNA damage sensor activity / single-stranded DNA binding / DNA recombination / damaged DNA binding / DNA repair / ATP binding / cytosol Similarity search - Function | ||||||
| Biological species |  Mycobacterium smegmatis str. MC2 155 (bacteria) Mycobacterium smegmatis str. MC2 155 (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.6 Å MOLECULAR REPLACEMENT / Resolution: 2.6 Å | ||||||
 Authors Authors | Prabu, J.R. / Manjunath, G.P. / Chandra, N.R. / Muniyappa, K. / Vijayan, M. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.D / Year: 2008 Journal: Acta Crystallogr.,Sect.D / Year: 2008Title: Functionally important movements in RecA molecules and filaments: studies involving mutation and environmental changes Authors: Prabu, J.R. / Manjunath, G.P. / Chandra, N.R. / Muniyappa, K. / Vijayan, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2zrj.cif.gz 2zrj.cif.gz | 77 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2zrj.ent.gz pdb2zrj.ent.gz | 55.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2zrj.json.gz 2zrj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zr/2zrj https://data.pdbj.org/pub/pdb/validation_reports/zr/2zrj ftp://data.pdbj.org/pub/pdb/validation_reports/zr/2zrj ftp://data.pdbj.org/pub/pdb/validation_reports/zr/2zrj | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2zr0C  2zr7C  2zr9C  2zraC  2zrbC  2zrcC  2zrdC  2zreC  2zrfC  2zrgC  2zrhC  2zriC  2zrkC  2zrlC  2zrmC  2zrnC  2zroC  2zrpC  1ubcS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 37287.441 Da / Num. of mol.: 1 / Mutation: Q196A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Mycobacterium smegmatis str. MC2 155 (bacteria) Mycobacterium smegmatis str. MC2 155 (bacteria)Plasmid: PTHIOA / Production host:  |
|---|
-Non-polymers , 5 types, 68 molecules 


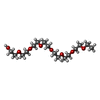





| #2: Chemical | ChemComp-AGS / | ||||
|---|---|---|---|---|---|
| #3: Chemical | ChemComp-CL / | ||||
| #4: Chemical | | #5: Chemical | ChemComp-PE4 / | #6: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.03 Å3/Da / Density % sol: 59.38 % |
|---|---|
| Crystal grow | Temperature: 296 K / Method: vapor diffusion, hanging drop / pH: 6.9 Details: 80mM citrate/phosphate buffer(pH 6.9), 80mM NaCl, 40mM ammonium acetate, 20mM sodium citrate, 6% poly-ethylene glycol 3350, 30% poly-ethylene glycol 3350, 200mM ammonium acetate in sodium ...Details: 80mM citrate/phosphate buffer(pH 6.9), 80mM NaCl, 40mM ammonium acetate, 20mM sodium citrate, 6% poly-ethylene glycol 3350, 30% poly-ethylene glycol 3350, 200mM ammonium acetate in sodium citrate buffer(PH 5.8), VAPOR DIFFUSION, HANGING DROP, temperature 296K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.54 Å ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.54 Å |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Nov 21, 2006 / Details: Mirrors |
| Radiation | Monochromator: Single crystal, cylindrically bent, Si(220) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 2.6→30 Å / Num. all: 13807 / Num. obs: 13807 / % possible obs: 100 % / Observed criterion σ(I): 0 / Redundancy: 4.8 % / Biso Wilson estimate: 52.3 Å2 / Rmerge(I) obs: 0.069 / Net I/σ(I): 18.5 |
| Reflection shell | Resolution: 2.6→2.74 Å / Redundancy: 4 % / Rmerge(I) obs: 0.371 / Mean I/σ(I) obs: 3 / Num. unique all: 1995 / % possible all: 99.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1UBC Resolution: 2.6→24.97 Å / Rfactor Rfree error: 0.007 / Data cutoff high absF: 1326391.39 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber / Details: BULK SOLVENT MODEL USED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 59.8464 Å2 / ksol: 0.35 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 57.4 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.6→24.97 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.6→2.76 Å / Rfactor Rfree error: 0.024 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj





