[English] 日本語
 Yorodumi
Yorodumi- PDB-2jw1: Structural characterization of the type III pilotin-secretin inte... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2jw1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structural characterization of the type III pilotin-secretin interaction in Shigella flexneri by NMR spectroscopy | ||||||
 Components Components |
| ||||||
 Keywords Keywords | MEMBRANE PROTEIN / protein-protein interaction / Lipoprotein / Membrane / Outer membrane / Palmitate / Plasmid / Virulence / Protein transport / Transport | ||||||
| Function / homology |  Function and homology information Function and homology informationtype III protein secretion system complex / type II protein secretion system complex / protein secretion by the type III secretion system / cell outer membrane Similarity search - Function | ||||||
| Biological species |  Shigella flexneri (bacteria) Shigella flexneri (bacteria)synthetic construct (others) | ||||||
| Method | SOLUTION NMR / simulated annealing, molecular dynamics, torsion angle dynamics | ||||||
 Authors Authors | Okon, M.S. / Lario, P.I. / Creagh, L. / Jung, Y.M.T. / Maurelli, A.T. / Strynadka, N.C.J. / McIntosh, L.P. | ||||||
 Citation Citation |  Journal: Structure / Year: 2008 Journal: Structure / Year: 2008Title: Structural Characterization of the Type-III Pilot-Secretin Complex from Shigella flexneri Authors: Okon, M. / Moraes, T.F. / Lario, P.I. / Creagh, A.L. / Haynes, C.A. / Strynadka, N.C. / McIntosh, L.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2jw1.cif.gz 2jw1.cif.gz | 679.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2jw1.ent.gz pdb2jw1.ent.gz | 572.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2jw1.json.gz 2jw1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jw/2jw1 https://data.pdbj.org/pub/pdb/validation_reports/jw/2jw1 ftp://data.pdbj.org/pub/pdb/validation_reports/jw/2jw1 ftp://data.pdbj.org/pub/pdb/validation_reports/jw/2jw1 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 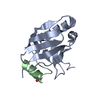
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 12903.888 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Shigella flexneri (bacteria) / Gene: mxiM / Production host: Shigella flexneri (bacteria) / Gene: mxiM / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 2131.315 Da / Num. of mol.: 1 / Fragment: sequence database residues 549-566 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) / References: UniProt: Q04641 |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: 0.3 -0.5 mM [U-95% 13C; U-95% 15N] MxiM(28-142), 0.5 - 0.7 mM MxiD(553-570), 95% H2O/5% D2O Solvent system: 95% H2O/5% D2O | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| ||||||||||||
| Sample conditions | Ionic strength: 20 / pH: 6.5 / Pressure: ambient / Temperature: 288 K |
-NMR measurement
| NMR spectrometer | Type: Varian INOVA / Manufacturer: Varian / Model: INOVA / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: simulated annealing, molecular dynamics, torsion angle dynamics Software ordinal: 1 Details: Additional water refinement was made for each of submitted conformers | ||||||||||||||||
| NMR representative | Selection criteria: closest to the average | ||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 200 / Conformers submitted total number: 15 |
 Movie
Movie Controller
Controller








 PDBj
PDBj





 HSQC
HSQC