[English] 日本語
 Yorodumi
Yorodumi- PDB-2hob: Crystal structure of SARS-CoV main protease with authentic N and ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2hob | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of SARS-CoV main protease with authentic N and C-termini in complex with a Michael acceptor N3 | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | VIRAL PROTEIN / SARS-CoV / main protease / michael acceptor N3 | |||||||||
| Function / homology |  Function and homology information Function and homology informationviral RNA-directed RNA polymerase complex / viral replication complex formation and maintenance / exoribonuclease complex / symbiont-mediated suppression of host TRAF-mediated signal transduction => GO:0039527 / : / : / : / cytoplasmic viral factory / positive regulation of ubiquitin-specific protease activity / symbiont-mediated suppression of host translation ...viral RNA-directed RNA polymerase complex / viral replication complex formation and maintenance / exoribonuclease complex / symbiont-mediated suppression of host TRAF-mediated signal transduction => GO:0039527 / : / : / : / cytoplasmic viral factory / positive regulation of ubiquitin-specific protease activity / symbiont-mediated suppression of host translation / : / : / endopeptidase complex / endoribonuclease complex / mRNA capping enzyme complex / positive stranded viral RNA replication / positive regulation of RNA biosynthetic process / Assembly of the SARS-CoV-1 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / Transcription of SARS-CoV-1 sgRNAs / protein K48-linked deubiquitination / Translation of Replicase and Assembly of the Replication Transcription Complex / K48-linked deubiquitinase activity / Replication of the SARS-CoV-1 genome / protein K63-linked deubiquitination / RNA-templated transcription / K63-linked deubiquitinase activity / host cell endoplasmic reticulum / viral transcription / protein autoprocessing / SARS-CoV-1 modulates host translation machinery / 7-methylguanosine mRNA capping / membrane => GO:0016020 / positive regulation of viral genome replication / Transferases; Transferring one-carbon groups; Methyltransferases / DNA helicase activity / helicase activity / protein processing / SARS-CoV-1 activates/modulates innate immune responses / double-stranded RNA binding / 5'-3' RNA helicase activity / Lyases; Phosphorus-oxygen lyases / endonuclease activity / ISG15-specific peptidase activity / Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters / double membrane vesicle viral factory outer membrane / SARS coronavirus main proteinase / host cell endoplasmic reticulum-Golgi intermediate compartment / host cell endosome / 3'-5'-RNA exonuclease activity / symbiont-mediated degradation of host mRNA / 5'-3' DNA helicase activity / mRNA guanylyltransferase / symbiont-mediated suppression of host toll-like receptor signaling pathway / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / mRNA guanylyltransferase activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / omega peptidase activity / mRNA (guanine-N7)-methyltransferase / methyltransferase cap1 / DNA helicase / symbiont-mediated suppression of host NF-kappaB cascade / host cell cytoplasm / symbiont-mediated perturbation of host ubiquitin-like protein modification / host cell Golgi apparatus / methyltransferase cap1 activity / cysteine-type deubiquitinase activity / mRNA 5'-cap (guanine-N7-)-methyltransferase activity / ubiquitinyl hydrolase 1 / Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases / lyase activity / protein dimerization activity / single-stranded RNA binding / viral protein processing / host cell perinuclear region of cytoplasm / RNA helicase / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / symbiont-mediated suppression of host gene expression / viral translational frameshifting / symbiont-mediated activation of host autophagy / RNA-directed RNA polymerase / cysteine-type endopeptidase activity / viral RNA genome replication / RNA-directed RNA polymerase activity / DNA-templated transcription / ATP hydrolysis activity / proteolysis / zinc ion binding / ATP binding / identical protein binding Similarity search - Function | |||||||||
| Biological species |  SARS coronavirus SARS coronavirussynthetic construct (others) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.95 Å MOLECULAR REPLACEMENT / Resolution: 1.95 Å | |||||||||
 Authors Authors | Xue, X. / Yang, H. / Shen, W. / Zhao, Q. / Li, J. / Rao, Z. | |||||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2007 Journal: J.Mol.Biol. / Year: 2007Title: Production of authentic SARS-CoV M(pro) with enhanced activity: application as a novel tag-cleavage endopeptidase for protein overproduction Authors: Xue, X. / Yang, H. / Shen, W. / Zhao, Q. / Li, J. / Yang, K. / Chen, C. / Jin, Y. / Bartlam, M. / Rao, Z. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2hob.cif.gz 2hob.cif.gz | 82.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2hob.ent.gz pdb2hob.ent.gz | 60.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2hob.json.gz 2hob.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ho/2hob https://data.pdbj.org/pub/pdb/validation_reports/ho/2hob ftp://data.pdbj.org/pub/pdb/validation_reports/ho/2hob ftp://data.pdbj.org/pub/pdb/validation_reports/ho/2hob | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2h2zC  1uk3S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
| ||||||||
| Details | The crystal belongs to space group C2, and each asymmetric unit contains only one protomer of a typical dimer. The two protomers in the dimer are related by the crystallographic 2-fold symmetry axis. |
- Components
Components
| #1: Protein | Mass: 33876.637 Da / Num. of mol.: 1 / Fragment: 3C-like proteinase Source method: isolated from a genetically manipulated source Source: (gene. exp.)  SARS coronavirus / Genus: Coronavirus / Plasmid: pGEX-6p-1 / Production host: SARS coronavirus / Genus: Coronavirus / Plasmid: pGEX-6p-1 / Production host:  References: UniProt: P59641, UniProt: P0C6X7*PLUS, Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases |
|---|---|
| #2: Protein/peptide | 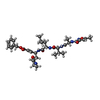  Type: Peptide-like / Class: Inhibitor / Mass: 680.791 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) Type: Peptide-like / Class: Inhibitor / Mass: 680.791 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others)References: N-[(5-METHYLISOXAZOL-3-YL)CARBONYL]ALANYL-L-VALYL-N~1~-((1R,2Z)-4-(BENZYLOXY)-4-OXO-1-{[(3R)-2-OXOPYRROLIDIN-3-YL]METHYL}BUT-2-ENYL)-L-LEUCINAMIDE |
| #3: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.36 Å3/Da / Density % sol: 63.35 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 6 Details: 2% polyethylene glycol(PEG) 6000, 3% DMSO, 1mM DTT, 0.1M [2-(N-morpholino)ethanesulfonic acid] (Mes) buffer (pH 6.0), with a protein concentration of 5mg/ml. Inhibitor N3 was dissolved in 7. ...Details: 2% polyethylene glycol(PEG) 6000, 3% DMSO, 1mM DTT, 0.1M [2-(N-morpholino)ethanesulfonic acid] (Mes) buffer (pH 6.0), with a protein concentration of 5mg/ml. Inhibitor N3 was dissolved in 7.5% PEG 6000, 6% DMSO, and 0.1M Mes (pH 6.0) with a concentration of 10mM (supersaturation). Then, a 3 micro-l aliquot of such solution was added to the drop, and the crystals were soaked for approximately 2-6 days., VAPOR DIFFUSION, HANGING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 Å ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 Å |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Jun 3, 2006 |
| Radiation | Monochromator: OSMIC MIRROR / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.95→50 Å / Num. obs: 32068 / % possible obs: 99.6 % |
| Reflection shell | Resolution: 1.95→2.02 Å / % possible all: 97.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1UK3 Resolution: 1.95→50 Å / σ(F): 0 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.95→50 Å
| ||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj








